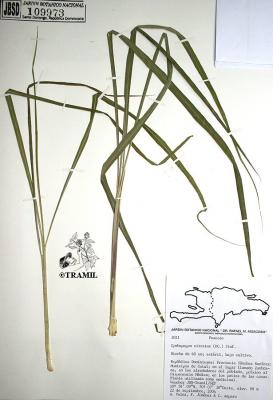
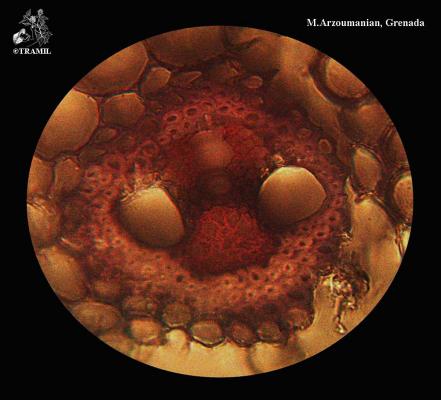
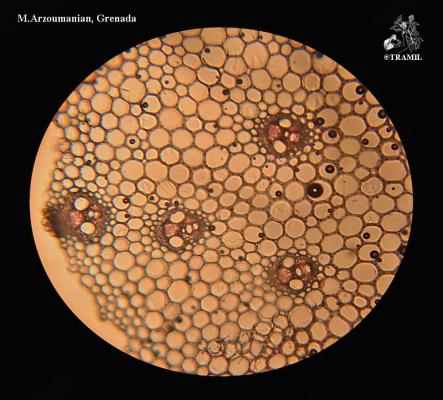
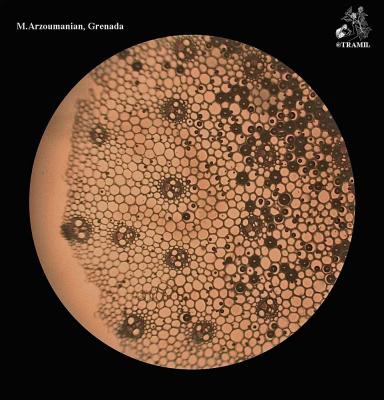
Cymbopogon citratus
Vernacular names
(In territories with significant traditional TRAMIL use)
Barbados:
- fever grass
- lemon grass
St Vincent and Grenadines:
- fever grass
- lemon grass
Antigua and Barbuda:
- fever grass
- lemon grass
Trinidad and Tobago:
- fever grass
- lemon grass
Puerto Rico:
- limoncillo
Dominican Republic:
- limoncillo
Venezuela:
- molojillo criollo
Guadeloupe:
- sitwonnèl
- zacate té
Dominica:
- sitwonnèl
- zacate té
Saint Martin:
- sitwonnèl
- zacate té
Martinique:
- sitwonnèl
- zacate té
Saint Lucia:
- sitwonnèl
- zacate té
Guatemala:
- té de limón
Honduras:
- té limón
- zacate limón
- zacate té
Mexico:
- té limón
- zacate limón
- zacate té
Costa Rica:
- té limón
- zacate limón
- zacate té
Other vernacular names
Cuba:
- Caña santa
Guadeloupe:
- Citronelle
- citronnelle
- Citronnelle (sitwonnèl)
Saint Martin:
- Citronnelle
Antigua and Barbuda:
- Fever grass
- Fever grass, lemon grass
Trinidad and Tobago:
- Fever grass = lemon grass
St Vincent and Grenadines:
- Fevergrass
Barbados:
- Lemon grass
Dominica:
- Lemon grass
Colombia:
- Lemon grass
- Limonaria
- Yerba santa
Dominican Republic:
- Limoncillo
Puerto Rico:
- Limoncillo
Venezuela:
- Malojillo
Haiti:
- Sitwonel
Martinique:
- sitwonnèl, citronnelle
Guatemala:
- Té de Limón
Nicaragua:
- Zacate de limón
Honduras:
- Zacate té
Geographical distribution
Native to India, cultivated in tropical and subtropical areas.
Botanical description
Perennial erect herb, 1 to 2 m, forming dense clusters. Leaves up to 1 m long, tapered towards both ends. Inflorescence up to 60 cm; segments of villous racemes; sessile spikelets, without bristles, linear to linear-lanceolate, flat on the back.
Voucher(s)
Faujour,9,BAR
Jean Pierre,273,SLNH
Ochoa,258,HPMVH
Girón,199&478,CFEH
García,2654,JBSD
Mejía,18,MAPR
Delens,264209-30,VEN
Balland,10,HVB
Delaigue,11,NHTT
Ríos,411,CECALLI
Boulogne,TH,10,UAG
Boulogne,TB,8,UAG
Picking,35474,UWI
Pharmacopoeia
Ed.3References
1 LAGOS-WITTE S, 1988-89, 1996
Encuesta TRAMIL. Laboratorio de Histología Vegetal y Etnobotánica, Dep. de Biología, Universidad Nacional Autónoma de Honduras UNAH, Tegucigalpa, Honduras.
2 WENIGER B, ROUZIER M, 1986
Enquête TRAMIL. Service Oecuménique d’Entraide SOE, Port au Prince, Haïti.
3 ZAMBRANO LE, 2007
Encuesta TRAMIL en Guareguare, Miranda. UCV, Caracas, Venezuela.
4 GIRON L, 1988
Encuesta TRAMIL (Costa atlántica). Centro Mesoamericano de Tecnología CEMAT, Guatemala, Guatemala.
5 CHARLES C, 1988
TRAMIL survey. Movement for Cultural Awareness MCA, Roseau, Dominica.
6 GERMOSEN-ROBINEAU L, GERONIMO M, AMPARO C, 1984
Encuesta TRAMIL. enda-caribe, Santo Domingo, Rep. Dominicana.
7 JEAN-PIERRE L, 1988
TRAMIL survey. St. Lucia National Herbarium, Castries, St. Lucia.
8 FAUJOUR A, MURREY D, CHELTENHAM-CORBIN B, CARRINGTON S, 2003
TRAMIL survey. enda-caribbean, IICA & UAG, Saint Thomas, Barbados.
9 BALLAND V, GLASGOW A, SPRINGER F, GAYMES G, 2004
TRAMIL survey. IICA, UAG & U.PARIS XI, Saint Vincent.
10 DELAIGUE J, 2005
TRAMIL survey. UAG & PRDI, Tobago House of Assembly, Scarborough, Tobago.
11 PICKING D, MITCHELL S, DELGODA R, YOUNGER N, 2011
TRAMIL survey. Natural Products Institute, The Biotechnology Centre & Tropical Metabolic Research Institute, University of the West Indies, Mona, Jamaica.
12 CLEMENT Y, BAKSH-COMEAU Y, RAGOO R, SEAFORTH C, 2008
TRAMIL survey. UWI & UTT, Trinidad.
13 OCAMPO R, 1988
Encuesta TRAMIL (Costa atlántica), Instituto de Desarrollo Agrario, Universidad de Costa Rica, San José, Costa Rica.
14 LONGUEFOSSE JL, NOSSIN E, 1990-95
Enquête TRAMIL. Association pour la valorisation des plantes médicinales de la Caraïbe AVPMC, Fort de France, Martinique.
15 DELENS M, 1992
Encuesta TRAMIL en los Estados Lara y Sucre de Venezuela. Centro al Servicio de la Acción Popular CESAP, Caracas, Venezuela.
16 OCRISSE G, 2008
Enquête TRAMIL auprès de 250 familles de la moitié Est de la partie francophone de St Martin. Biologie végétale, UAG, Guadeloupe.
17 BOULOGNE I, 2009
Enquête TRAMIL, (Terre-de-Bas et Terre-de-Haut) Les Saintes, UAG, Guadeloupe.
18 O’REILLY A, 1992
TRAMIL survey. Chemistry & Food Technology Division, Ministry of Agriculture, Dunbars, Antigua & Barbuda.
19 BENEDETTI MD, 1994
Encuesta TRAMIL. Universidad de Puerto Rico, Mayagüez, Puerto Rico.
20 BALZ E, BOYER A, BURAUD M, 2007
Enquête TRAMIL à Marie-Galante. U. Bordeaux 3, U. Paris XI Chatenay-Malabry, UAG, Guadeloupe.
21 BOYER A, BURAUD M, 2007
Enquête TRAMIL à La Désirade. U. Paris XI Chatenay-Malabry, UAG, Guadeloupe.
22 MENDEZ M, MEDINA ML, DURAN R, 1996
Encuesta TRAMIL. Unidad de recursos naturales, Centro de Investigación Científica de Yucatán CICY, Mérida, México.
23 ABEGAZ B, YOHANNES P, DIETER R, 1983
Constituents of the essential oil of Ethiopian Cymbopogon citratus. J Nat Prod 46(3):424-426.
24 DE MATOUSCHEK B, STAHL-BISKUP E, 1991
Phytochemical investigation of nonvolatile constituents of Cymbopogon citratus (DC.) Stapf. (Poaceae). Pharm Acta Helv 66(9/10):242-245.
25 HANSON S, CRAWFORD M, KOKER M, MENEZES F, 1976
Cymbopogonol, a new triterpenoid from Cymbopogon citratus. Phytochemistry 15:1074-1075.
26 YOKOYAMA Y, TSUYUKI T, NAKAMURA N, TAKAHASHI T, HANSON S, MATSUSHITA K, 1980
Revised structures of cymbopogone and cymbopogonol. Tetrahedron Lett 21:3701-3702.
27 OLANIYI A, SOFOWORA E, OGUNTIMEHIN B, 1975
Phytochemical investigation of some Nigerian plants used against fevers. II. Cymbopogon citratus. Planta Med 28:186-189.
28 DUKE JA, ATCHLEY AA, 1986
Handbook of proximate analysis tables of higher plants. Boca Raton, USA: CRC Press. p57.
29 SAUVAIN M, MORETTI C, MUÑOZ V, 1990
Pruebas in vivo para paludismo realizadas en Bolivia sobre varias plantas TRAMIL. ORSTOM/IRD/IBBA, La Paz, Bolivia.
30 MORON F, SANCHEZ C, MARTINEZ MC, MOREJON Z, PINEDO Z, 2000
Actividad antiespasmódica in vitro de hojas frescas de Cymbopogon citratus (DC.) Stapf. Informe TRAMIL. Laboratorio Central de Farmacología, Facultad de Ciencias Médicas “Dr. Salvador Allende”, La Habana, Cuba.
31 MORON F, FURONES J, PINEDO Z, 1996
Ausencia de efectos antiinflamatorio y analgésico del extracto fluído de Cymbopogon citratus al 30% por vía oral. Rev Cubana Plant Med 1(2):3-6.
32 CARBALLO A, 1995
Plantas medicinales del Escambray cubano. Informe TRAMIL. Laboratorio provincial de producción de medicamentos, Sancti Spiritus, Cuba.
33 WENIGER B, ROUZIER M, DAGUILH R, HENRYS D, HENRYS J, ANTON R, 1986
Popular medicine of the central plateau of Haiti. 2. Ethnopharmacological inventory. J Ethnopharmacol 17(1):13-30.
34 CARLINI EA, CONTAR JD, SILVA-FILHO AR, SOLVEIRA FILHO NG, FROCHTENGARTEN ML, BUENO OF, 1986
Pharmacology of lemon-grass Cymbopogon citratus I. Effect of teas prepared from the leaves on laboratory animals. J Ethnopharmacol 17(1):37-64.
35 SOUZA FORMIGONI ML, LODDER HM, FILHO OG,FERREIRA TM, CARLINI EA, 1986
Pharmacology of lemongrass (Cymbopogon citratus Stapf). II. Effects of daily two month administration in male and female rats and in offspring exposed “in utero”. J Ethnopharmacol 17(1):65 74.
36 CARBAJAL D, CASACO A, ARRUZAZABALA L, GONZALEZ R, TOLON Z, 1989
Pharmacological study of Cymbopogon citratus leaves. J Ethnopharmacol 25(1):103-107.
37 LAM L, ZHENG B, 1991
Effects of essential oils on glutathione S-transferase activity in mice. J Agric Food Chem 39(4):660-662.
38 LORENZETTI B, SOUZA G, SARTI S, FILHO DS, FERREIRA SH, 1991
Myrcene mimics the peripheral analgesic activity of lemongrass tea. J Ethnopharmacol 34(1):43-48.
39 SETH, G, KOKATE CK, VARMA KC, 1976
Effect of essential oil of Cymbopogon citratus on central nervous system. Indian J Exp Biol 14(3):370-371.
40 LEMOS TLG, MATOS FJA, ALENCAR JW, CRAVEIRO AA, CLARK AM, MC CHESNEY JD, 1990
Antimicrobial activity of essential oils of Brazilian plants. Phytother Res 4(2):82-84.
41 AWUAH R, 1989
Fungitoxic effects of extracts from some West African plants. Ann Appl Biol 115(3):451-453.
42 DUKE JA, 1992
Handbook of biologically active phytochemicals and their bioactivities. Boca Raton, USA: CRC Press.
43 KOBAYASHI N, 1989
Pharmaceutical compositions containing lemongrass extracts and antioxidants. Patens Japan Kokai Tokio Koho., 01, 221, 320.
44 MARTINEZ MJ, BETANCOURT J, LOPEZ M, MOREJON Z, BARCELO H, LAINEZ A, MONTES ME, REGO R, BOUCOURT E, MORON F, 2000
Toxicidad aguda clásica de hoja seca de Cymbopogon citratus (DC.) Stapf. Informe TRAMIL. Laboratorio Central de Farmacología, Facultad de Ciencias Médicas “Dr. Salvador Allende”, La Habana, Cuba.
45 MARTINEZ MJ, BETANCOURT J, LOPEZ M, MOREJON Z, BOUCOURT E, MORON F, 2000
Actividad genotóxica in vitro de hoja seca de Cymbopogon citratus (DC.) Stapf. Informe TRAMIL. Laboratorio Central de Farmacología, Facultad de Ciencias Médicas “Dr. Salvador Allende”, La Habana, Cuba.
46 DE LA TORRE RA, ESPINOSA-AGUIRRE JJ, CORTINAS DE NAVA C, IZQUIERDO T, MORON F, 1994
Genotoxic activity of mebendazole in Aspergillus nidulans. Mutat Res 305(2):139-144.
47 PILOTO FERRER J, VIZOSO A, RAMOS A, GARCIA A, REMIGIO A, VEGA Y, GONZALEZ ML, RODRIGUEZ C, CARBALLO C, 2009
Plantas medicinales. Diez años de evaluaciones toxicogenéticas en el CIDEM. Boletín Latinoamericano y del Caribe de Plantas Medicinales y Aromáticas, 8(5):428-434.
48 LEITE JR, SEABRA ML, MALUF E, ASSOLANT K, SUCHECKI D, TUFIK S, KLEPACZ S, CALIL HM, CARLINI EA, 1986
Pharmacology of lemongrass (Cymbopogon citrates Stapf). III. Assessment of eventual toxic, hypnotic and anxiolytic effects on humans. J Ethnopharmacol 17(1):75-83.
49 PARRA AL, YHEBRA RS, SARDINAS IG, BUELA LI, 2001
Comparative study of assay of Artemia salina L. and the estimate of the medium lethal dose (LD50 value) in mice, to determine oral acute toxicity of plant extracts. Phytomedicine 8(5):395-400.
50 CARBALLO A, 1995
Cálculo de concentración y dosis de las drogas vegetales TRAMIL: Mensuraciones farmacognósticas y aproximaciones técnico-clínicas. Laboratorio provincial de producción de medicamentos, Sancti Spiritus, Cuba.