Crescentia cujete
Vernacular names
(In territories with significant traditional TRAMIL use)
Dominican Republic:
- higüero, güira
Geographical distribution
Native of continental tropical America, cultivated and naturalised in the tropics.
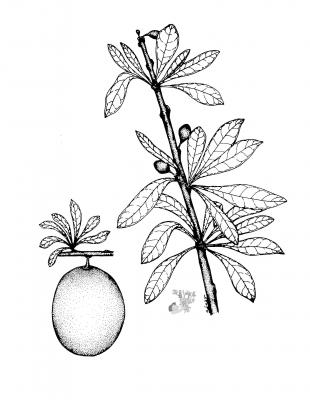
Botanical description
Tree 4-5 (rarely 8) meters high, trunk up to 20 cm in diameter and extended branches. Spatulate, fasciculate, short petiolate leaves, from 5 to 20 cm. Single, yellowish flowers with purple veins, 5 to 6 cm; lacerate lobes. Globose or ellipsoid fruit, smooth, glossy, 10 to 30 cm in diameter, hard shell.
Voucher(s)
Jiménez,22,JBSD
Slane,974,SLNH
Pharmacopoeia
Ed.3References
1 GERMOSEN-ROBINEAU L, GERONIMO M, AMPARO C, 1984
Encuesta TRAMIL. enda-caribe, Santo Domingo, Rep. Dominicana.
2 JOSEPH H, BOURGEOIS P, 1989
Contribution à la connaissance de Crescentia cujete L. Rapport TRAMIL. Laboratoire de phytochimie, Université des Antilles et de la Guyane, Pointe à Pitre, Guadeloupe.
3 ZWAVING J, 1986
Selección fitoquímica preliminar en algunas plantas TRAMIL. Laboratorio de Farmacognosia, Universidad de Groningen, Groningen, Nederland.
4 AGARWAL K, POPLI SP, 1992
The constituents of Crescentia cujete leaves. Fitoterapia 63(5):476.
5 GRIFFITHS LA, 1959
On the distribution of gentisic acid in green plants. J Exp Biol 10:437.
6 LE GRAND A, WONDERGEM PA, 1986
Antimicrobial activity of 10 Caribbean species. TRAMIL inform. Dep. of Pharmacognosy, University of Leyden, Leyden, Nederland.
7 GUPTA M, ESPOSITO AVELLA M, 1988
Evaluación química y farmacológica de algunas plantas medicinales de TRAMIL. CIFLORPAN, Universidad de Panamá, Panamá, Panamá.
8 VERPOORTE R, DIHAL PP, 1987
Medicinal plants of Surinam. IV. Antimicrobial activity of some medicinal plants. J Ethnopharmacol 21(3):315-318.
9 CONTRERAS A, ZOLLA C, 1982
Plantas tóxicas de México. Instituto Mexicano del Seguro Social, México DF, México.
10 LOPEZ M, GARCIA AI, BOUCOURT E, BACALLAO Y, MORON F, 2012
Ausencia de irritabilidad dérmica primaria sobre piel sana y dañada del zumo de la hoja fresca calentada de Cresencia crujete. Trabajo TRAMIL. Laboratorio Central de Farmacología. Facultad de C. Médicas “Dr. Salvador Allende”. La Habana.
11 CACERES A, Ed., 2005
Vademecum de plantas de uso medicinal en Guatemala. Facultad de Ciencias Químicas y Farmacia, Universidad de San Carlos, Guatemala, Guatemala.