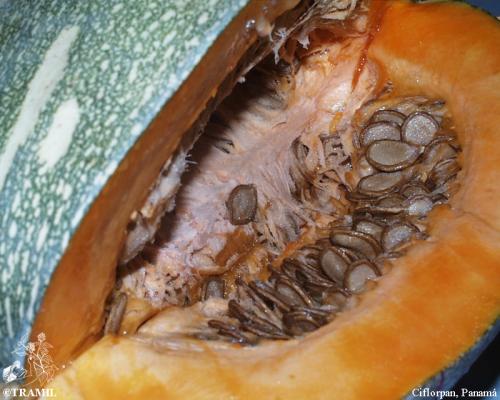
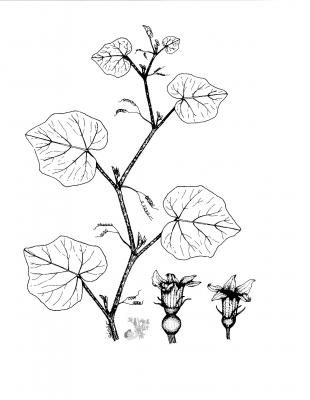
Cucurbita moschata
Vernacular names
(In territories with significant traditional TRAMIL use)
Dominican Republic:
- auyama
Haiti:
- jiroumou
- jiwomon
Geographical distribution
Native to tropical America, widely cultivated in tropical and subtropical regions.
Botanical description
Herbaceous annual, creeping or ascending, with slightly angular stems. Simple or ramified tendrils. Pubescent, serrate leaves with 5 to 7 acute or obtuse lobes. Unisexual flowers, axillary,
solitary; orange-yellow corolla of up to 15 cm. Globose fruit, sometimes cylindrical, pyriform or conical, green, yellow or orange, often grooved.
Voucher(s)
Jiménez,127,JBSD
Pharmacopoeia
Ed.3References
1 WENIGER B, ROUZIER M, 1986
Enquête TRAMIL. Service Oecuménique d’Entraide SOE, Port au Prince, Haïti.
2 WENIGER B, 1987-88
Encuesta TRAMIL. enda-caribe, Santo Domingo, Rep. Dominicana.
3 HEGNAUER R, 1973
Chemotaxonomy der Pflanzen. Basel, Schweiz: Birkhauser Verlag. 6:882.
4 WASHUTTL J, REIDERER P, BANCHER E, 1973
A qualitative and quantitative study of sugar-alcohols in several foods. J Food Sci 38(7):1262-1263.
5 DUKE JA, 2000
Chemicals and their Biological Activities in: Cucurbita moschata. Dr. Duke’s Phytochemical and Ethnobotanical Databases. USDA-ARS-NGRL, Beltsville Agricultural Research Center, Beltsville, USA, August 10, 2000.www.arsgrin.gov/cgi-bin/duke/farmacy2.pl
6 BANG MH, HAN JT, KIM HY, PARK YD, PARK CH, LEE KR, BAEK NI, 2002
13-Hydroxy-9Z,11E,15E-octadecatrienoic acid from the leaves of Cucurbita moschata. Arch Pharm Res
25(4):438-440.
7 DUKE JA, ATCHLEY AA, 1986
Handbook of proximate analysis tables of higher plants. Boca Raton, USA: CRC Press. p55.
8 USDA.
www.nal.usda.gov/fnic/cgi-bin/nut_search.pl?squash
9 HERRERA J, 1992
Determinación de parámetros farmacológicos usados en medicina tradicional popular en la cuenca del Caribe. Informe TRAMIL. Laboratorio de Fitofarmacología, Dep. de Farmacología, Facultad de Salud, Universidad del Valle, Cali, Colombia.
10 PAZOS L, COTO T, CAIZA F, 2008
Antiinflamatorio tópico, en ratones, de la hoja de Curcubita moschata. Informe TRAMIL, Laboratorio de
Ensayos Biológicos, LEBi, Universidad de Costa Rica, San Pedro, Costa Rica.
11 VILENCHIK M, 1989
Fundamentos biológicos del envejecimiento y la longevidad. Moscú, URSS: Ed. MIR.
12 HERRERA J, 1990
Determinación de parámetros farmacológicos de vegetales utilizados en medicina tradicional en la cuenca del Caribe. Informe TRAMIL. Laboratorio de Fitofarmacología, Dep. de Farmacología, Facultad de Salud,
Universidad del Valle, Cali, Colombia.
13 WENIGER B, 1992
Cytotoxicité, effets immunodulateurs et morphologique des extraits éthanolique 80% et aqueux de feuille de
Cucurbita. Rapport TRAMIL. Faculté de Pharmacie, Université de Strasbourg, Illkirch, France.
14 HURTADO M, CARBALLO A, 1990
Las plantas medicinales TRAMIL en la farmacopea Soviética. Centro de Investigaciones de Fitoterapia y
Medicina Tradicional, Topes de Collantes, Cuba.
15 PARIS R, MOYSE H, 1981
Précis de matière médicale. Paris, France: Ed. Maloine.
16 CHEN ZK, PU TC, LI DY, JIANG HA, 1980
Therapeutic effect of cucurbitine on dog taeniasis. Zhongguo Yao Li Xue Bao 1(2):124-126.
17 ALBORNOZ A, 1993
Medicina tradicional herbaria. Guía de Fitoterapia. Caracas, Venezuela: Editorial Instituto Farmacoterápico
Latino S.A. p226.