Vernacular names
(In territories with significant traditional TRAMIL use)
Dominican Republic:
- café
Haiti:
- kafé
Geographical distribution
Native to Africa, cultivated in tropical regions.
Botanical description
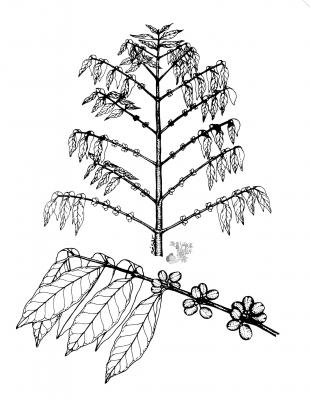
Shrub up to 7 m high, thin trunk, straight. Short-petiolate leaves, elliptic to oblong, shiny, subcoriaceous, from 7 to 15 cm. Flowers in axillary groups, fragrant, usually sessile; truncated calyx; white petals. Oblong to globose berry, 10 to 16 mm, red when ripe.
Voucher(s)
Jiménez,122,JBSD
Volcius,165,SOE
Pharmacopoeia
Ed.3References
1 WENIGER B, ROUZIER M, 1986
Enquête TRAMIL. Service Oecuménique d'Entraide SOE, Port au Prince, Haïti.
2 GERMOSEN-ROBINEAU L, GERONIMO M, AMPARO C, 1984
Encuesta TRAMIL. enda-caribe, Santo Domingo, Rep. Dominicana.
3 WENIGER B, 1987-88
Encuesta TRAMIL. enda-caribe, Santo Domingo, Rep. Dominicana.
4 QUILEZ AM, GARCIA D, SAENZ T, 2009
Uso racional de medicamentos a base de plantas. Guía de interacciones entre fitomedicamentos y fármacos de síntesis. Sevilla, España: 1a Edición Fundación Farmacéutica Avenzoar.
5 MIYAKE T, SHIBAMOTO T, 1993
Quantitative analysis of acetaldehyde in foods and beverages. J Agric Food Chem 41(11):1968-1970.
6 KASAI H, KUMENO K, AMAIZUMI Z, NISHIMURA S, NAGAO M, FUJITA Y, SUGIMURA T, NUKAYA H, KOSUGE T, 1982
Mutagenicity of methylglyoxal in coffee. Jpn J Cancer Res (GANN) 73:681-683.
7 NEURATH GB, DUNGER M, PEIN FG, AMBROSIUS D, SCHREIBER O, 1977
Primary and secondary amines in the human environment. Food Cosmet Toxicol 15(4):275-282.
8 AMORIM HV, CORTEZ JG, 1973
Methods of organic analysis of coffee. II. Comparison of methods of caffeine determination in green coffee. An Esc Super Agr Luiz De Queiroz Univ Sao Paulo 30:281.
9 DEISINGER PJ, HILL TS, ENGLISH JC, 1996
Human exposure to naturally occurring hydroquinone. J Toxicol Environ Health 47(1):31-46.
10 NISHINA A, KAJISHIMA F, MATSUNAGA M, TEZUKA H, INATOMI H, OSAWA T, 1994
Antimicrobial substance, 3',4'-dihydroxyacetophenone, in coffee residue. Biosci Biotechnol Biochem 58(2):293-296.
11 AESCHBACH R, KUSY A, MAIER HG, 1982
Diterpenes of coffee. I. Atractyligenin. Z Lebensm-Unters Forsch 175(5):337-341.
12 GROSS G, JACCAUD E, HUGGETT AC, 1997
Analysis of the content of the diterpenes cafestol and kahweol in coffee brews. Food Chem Toxicol 35(6):547-554.
13 DUPLATRE A, TISSE C, ESTIENNE J, 1984
Identification of arabica and robusta [coffee] species by studying the sterol fraction. Ann Falsif Expert Chim Toxicol 77(828):259-270.
14 ANDRADE PB, LEITAO R, SEABRA RM, OLIVEIRA MB, FERREIRA MA, 1997
Development of an HPLC/diode-array detector method for simultaneous determination of seven hydroxy-cinnamic acids in green coffee. J Liq Chromatogr Relat Technol 20(13):2023-2030.
15 SONDHEIMER E, 1958
On the distribution of caffeic acid and the chlorogenic acid isomers in plants. Arch Biochem Biophys 74(1):131-138.
16 MEISSNER W, PODKOWINSKA H, WALKOWSKI A, 1974
Determination of chlorogenic acids in green coffee. Zesz Nauk Akad Ekon Poznaniu Ser 1(58):71.
17 OKUDA T, HATANO T, AGATA I, NISHIBE S, KIMURA K, 1986
Tannins in Artemisia montana, A. princeps and related species of plant. Yakugaku Zasshi 106(10):894-899.
18 HAGGAG MY, 1975
A study of the lipid content of Coffea arabica L. seeds. Pharmazie 30(6):409.
19 MAZAAFERA P, 1991
Trigonelline in coffee. Phytochemistry 30(7):2309-2310.
20 TSUJI S, SHIBATA T, OHARA K, OKADA N, ITO Y, 1991
Factors affecting the formation of hydrogen peroxide in coffee. Shokuhin Eiseigaku Zasshi 32(6):504-512.
21 STOFFELSMA J, SIPMA G, KETTENES DK, PYPKER J, 1968
New volatile components of roasted coffee. J Agric Food Chem 16(6):1000.
22 SPIRO M, 1997
Coffee, tea and chemistry. Chem Rev 6(5):11-15.
23 KOENIG WA, RAHN W, VETTER R, 1980
Identify and quantify emetic active constituents in roast coffee. Colloq Sci Int Café [C.R.] 9:145-149.
24 HOFMANN E, SCHLEE D, REINBOTHE H, 1969
On the occurrence and distribution of allantoin in Boraginaceae. Flora Abt A Physiol Biochem (Jena) 159:510-518.
25 MOLINA MR, DE LA FUENTE G, BATTEN MA, BRESSANI R, 1974
Decaffeination. A process to detoxify coffee pulp. J Agric Food Chem 22(6):1055.
26 KOLLING-SPEER I, SPEER K, 1997
Diterpenes in coffee leaves. Colloq Sci Int Café [C.R.] 17(15):1-154.
27 WALLER GR, JURZYSTE M, KARNS TKB, GENO PW, 1991
Isolation and identification of ursolic acid from Coffea arabica L. (coffee) leaves. Colloq Sci Int Cafe [C.R.] 14:245-247.
28 HIGUCHI K, SUZUKI T, ASHIHARA H, 1995
Pipecolic acid from the developing fruits (pericarp and seeds) of Coffea arabica and Camellia sinensis. Colloq Sci Int Café [C.R.] 16:389-395.
29 GONZALEZ J, NORIEGA R, SANDOVAL R, 1975
Contribution to the study of flavonoids of coffee tree (Coffea) leaves. Rev Colomb Quim 5:85.
30 CHOU C, WALLER G, 1980
Isolation and identification by mass spectrometry of phytotoxins in Coffea arabica. Bot Bull Acad Sinica (Taiwan) 21(1):25-34.
31 SERAFIN WE, 1996
Drugs used in the treatment of asthma. In: Hardman JG, Gilman AG, Limbird LE Eds. Goodman & Gilman’s The Pharmacological Basis of Therapeutics. 9th ed. New York, USA: The McGraw-Hill Professional Publishing, International Edition. p672-679.
32 DUKE JA, ATCHLEY AA, 1986
Handbook of proximate analysis tables of higher plants. Boca Raton, USA: CRC Press. p47.
33 CAMBAR P, CANALES M, CASTRO E, CASTRO C, MEJIA A, MEDINA F, LAGOS K, AGUILAR J, 1996
Efectos respiratorios y cardiovasculares de los extractos acuosos de las hojas de Coffea arabica L. en conejos. Informe TRAMIL. Unidad de Farmacología, Facultad de Ciencias Médicas, Universidad Nacional Autónoma de Honduras UNAH, Tegucigalpa, Honduras.
34 GARCIA M, Coto MT, González CS, Pazos L, 1998
Actividad bronquial del extracto acuoso de hoja fresca de Coffea arabica. Informe TRAMIL. Laboratorio de Ensayos Biológicos LEBi, Escuela de Medicina, Universidad de Costa Rica, San Pedro, Costa Rica.
35 CAMBAR P, CANALES M, GAMES V, CASTRO E, MEJIA A, CASTRO C, 1996
Efectos de los extractos acuosos de las hojas de Coffea arabica L. en la producción de úlceras gástricas por ligadura del píloro en ratas. Informe TRAMIL. Unidad de Farmacología, Facultad de Ciencias Médicas, Universidad Nacional Autónoma de Honduras UNAH, Tegucigalpa, Honduras.
36 DAGLIA M, CUZZONI MT, DACARRO C, 1994
Antibacterial activity of coffee. J Agric Food Chem 42(10):2270-2272.
37 TODA M, OKUBO S, HIYOSHI R, SHIMAMURA T, 1989
The bactericidal activity of tea and coffee. Lett Appl Microbiol 8(4):123-125.
38 KOLEY J, KOLEY BN, MAITRA SR, 1973
Effect of drinking tea, coffee and caffeine on work performance. Indian J Physiol Allied Sci 27:96.
39 ESTLE C, 1982
Caffeine psychotrope agents. Berlin, Germany: Springer verlag, 17:369-389.
40 CURATOLO PW, ROBERTSON D, 1983
The health consequences of caffeine. Ann Intern Med 98(5):641-653.
41 GREDEN JF, 1974
Anxiety of caffeinism. A diagnostic dilemma. Am J Psychiatry 131:1089-1092.
42 REY H, 1979
Effet d'un gel de caféine par voie cutanée sur la lipolyse locale. Thèse doctorat Médecine, Bordeaux, France.
43 THIERMAM-DUFFAUD D, 1983
Le café augmente-t-il la cholestérolémie? La Presse Médicale 12(34):2062.
44 DEBAS HT, COHEN MM, HOLUBITSKY IB, HARRISON RC, 1971
Caffeine simulated gastric and pepsine secretion: dose-response studies. Scand J Gastroenterol 6(5):453-457.
45 WRIGHT LF, GIBSON RG, HIRSCHOWITZ RI, 1977
Lack of caffeine stimulation of gastric secretion release in man. Proc Soc Exp Biol Med 154(4):538-539.
46 MCARTHUR K, HOGAN D, ISENBERG JI, 1982
Relative stimulatory effects of commonly ingested beverages on gastric secretion in human. Gastroenterology 83(1/2):199-203.
47 GARCIA M, Coto MT, González CS, Pazos L, 1998
Toxicidad aguda en ratones, del extracto acuoso de hojas frescas de Coffea arabica. Informe TRAMIL. Laboratorio de Ensayos Biológicos LEBI, Escuela de Medicina, Universidad de Costa Rica, San Pedro, Costa Rica.
48 STADLER RH, TURESKY RJ, MULLER O, MARKOVIC J, LEONG-MORGENTHALER PM, 1994
The inhibitory effects of coffee on radical-mediated oxidation and mutagenicity. Mutat Res 308(2):177-190.
49 STICH HF, ROSIN MP, BRYSON L, 1982
Inhibition of mutagenicity of a model nitrosation reaction by naturally occurring phenolics, coffee and tea. Mutat Res 95(2-3):119-128.
50 OBANA H, NAKAMURA SI, TANAKA RI, 1986
Suppressive effects of coffee on the SOS responses induced by UV and chemical mutagens. Mutat Res 175(2):47-50.
51 FUJITA FY, WAKABAYASHI K, NAGAO M, SUGIMURA T, 1985
Characteristics of major mutagenicity of instant coffee. Mutat Res 142(4):145-148.
52 WURZNER HP, LINDSTROM E, VUATAZ L, LUGINBUHL H, 1977
A 2-year feeding study of instant coffees in rats. I. Body weight, food comsumption, hematological parameters and plasma chemistry. Food Cosmet Toxicol 15(1):7-16.
53 NOLEN GA, 1981
The effect of brewed and instant coffee on reproduction and teratogenesis in the rat. Toxicol Appl Pharmacol 58(2):171-183.
54 ABRAHAM SK, 1995
Inhibitory effects of coffee on transplacental genotoxicity in mice. Mutat Res 347(1):45-52.
55 WURZNER HP, LINDSTROM E, VUATAZ L, LUGINBUHL H, 1977
A 2-year feeding study of instant coffees in rats. II. Incidence and types of neoplasms. Food Cosmet Toxicol 15(4):289-296.
56 MURPHY SJ, BENJAMIN CP, 1981
The effects of coffee on mouse development. Microbiol Lett 17:91-100.
57 NAGASAWA H, YASUDA M, SAKAMOTO S, INATOMI H, 1995
Protection by coffee cherry against spontaneous mammary tumour development in mice. Anticancer Res 15(1):141-146.
58 HASEGAWA R, ITO N, 1992
Liver medium-term bioassay in rats for screening of carcinogens and modifying factors in hepatocarcinogenesis. Food Chem Toxicol 30(11):979-992.
59 KLEEMOLA P, JOUSILAHTI P, PIETINEN P, VARTIAINEN E, TUOMILEHTO J, 2000
Coffee consumption and the risk of coronary heart disease and death. Arch Intern Med 160(22):3393-3400.
60 WILLETT WC, STAMPFER MJ, MANSON JE, COLDITZ GA, ROSNER BA, SPEIZER RE, HENNEKENS CH, 1996
Coffee consumption and coronary heart disease in women. J Amer Med Assoc 275(6):458-462.
61 TAVANI A, PREGNOLATO A, LA VECCHIA C, NEGRI E, TALAMINI R, FRANCESCHI S, 1997
Coffee and tea intake and risk of cancers of the colon and rectum: a study of 3,530 cases and 7,057 controls. Int J Cancer 73(2):193-197.
62 BARON JA, GREENBERG ER, HAILE R, MANDEL J, SANDLER RS, MOTT L, 1997
Coffee and tea and the risk of recurrent colorectal adenomas. Cancer Epidemiol Biomarkers Prev 6(1):7-10.
63 LUBIN F, RON E, WAX Y, MODAN B, 1985
Coffee and methylxanthines and breast cancer: a case-control study. J Natl Cancer Inst 74(3):569-573.
64 WILLIAMS MA, MONSON RR, GOLDMAN MB, MITTENDORF R, 1990
Coffee and delayed conception. Lancet 335(8705):1603.
65 PIRACCINI BM, BARDAZZI F, VINCENZI C, TARDIO MP, 1990
Occupational contact dermatitis due to coffee. Contact Dermatitis 23(2):114.
66 NISHIBE Y, TOMONO N, HIRASAWA H, OKADA T, 1996
Skin-lightening cosmetics containing extracts of Coffea arabica seeds. Patent-Japan Kokai Tokkyo Koho-08 92,057.