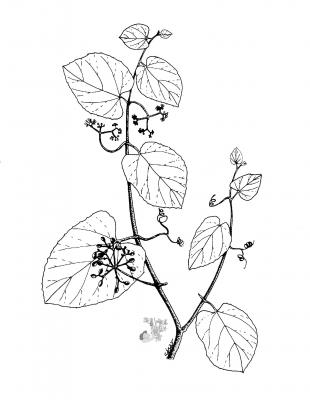
Cissus verticillata
Vernacular names
(In territories with significant traditional TRAMIL use)
Dominican Republic:
- bejuco caro
Haiti:
- lyann mòl
Geographical distribution
Tropical regions of the American continent.
Botanical description
Climbing plant, glabrous with tendrils; articulated branches. Alternate, simple, oblong to ovate leaves, up to 15 cm, densely pilose to glabrous, with a slightly serrated margin. Pedunculate cymes, yellow, highly ramified; small, green or yellow-green flowers. Ovoid or globose berries, purple to black, 8 to 10 mm, with a single seed.
Voucher(s)
García, 2326, JBSD
Cénesca, 201&202, SOE
Pharmacopoeia
Ed.3References
1 WENIGER B, ROUZIER M, 1986
Enquête TRAMIL. Service Oecuménique d’Entraide SOE, Port au Prince, Haïti.
2 GERMOSEN-ROBINEAU L, GERONIMO M, AMPARO C, 1984
Encuesta TRAMIL. enda-caribe, Santo Domingo, Rep. Dominicana.
3 WENIGER B, SAVARY H, DAGUIHL R, 1984
Tri phytochimique de plantes de la liste TRAMIL. Laboratoire de chimie des substances naturelles, Faculté de Médecine et de Pharmacie, Université d’Etat d’Haïti, Port au Prince, Haïti.
4 PINEDA M, 1990
Fitoquímica de algunas plantas TRAMIL. Laboratorio enda-caribe, Santo Domingo, Rep. Dominicana.
5 GARCIA MD, QUILEZ AM, SAENZ MT, MARTINEZ-DOMINGUEZ ME, DE LA PUERTA R, 2000
Anti-inflammatory activity of Agave intermixta Trel. and Cissus sicyoides L., species used in the Caribbean traditional medicine. J Ethnopharmacol 71:395-400.
6 BELTRAME FL, SARTORETTO JL, BAZOTTE RB, CUMAN RN, CORTEZ DAG, 2001
Estudo fitoquímico e avaliação do efeito antidiabético do Cissus verticillata L (Vitaceae). Química Nova 24:783-785.
7 BARBOSA WLR, SANTOS WRAS, PINTO LN, TAVARES ICC, 2002
Flavonóides de Cissus verticillata e a atividade hipoglicemiante do chá de suas folhas. Rev Bras Farmacognosia 12:13-15.
8 DE SOUSA C, DE PAIVA T, BEZERRA P, FALCÃO J, OLIVEIRA F, SILVEIRA E, FERREIRA J, FREIRE D, RODRIGUES M, CLÉA F, DE SOUSA F, DE CASTRO D, DA ROCHA S, DE BARROS G, 2007
Anti-diabetic activity of a fraction from Cissus verticillata and tyramine, its main bioactive constituent, in alloxan-induced diabetic rats. American J of Pharmacology and Toxicology 2(4):178-188.
9 STANDLEY PC, 1937
The flora of Costa Rica, Pt. I. Bot Serv Field Mus Nat Hist 18(391):398.
10 MARTINEZ MJ, LOPEZ M, BOUCOURT E, FUENTES V, MORON F, 2002
Evaluación de la actividad antiestafilocóccica de Cissus verticillata. Informe TRAMIL. Laboratorio Central de Farmacología, Facultad de Ciencias Médicas “Dr. Salvador Allende”, La Habana, Cuba.
11 LE GRAND A, WONDERGEM PA, 1986
Activités antimicrobiennes et études bibliographiques de la toxicologie de dix plantes médicinales de la Caraïbe. Rapport TRAMIL. Dép. de Pharmacognosie, Universités de Groningen & Leyden, Hollande.
12 FENG PC, HAYNES LJ, MAGNUS KE, PLIMMER JR, 1964
Pharmacological screening of some West Indian medicinal plants. J Pharm Pharmacol 16:115-117.
13 JIU J, 1966
A survey of some medicinal plants of Mexico for selected biological activities. Lloydia 29:250-259.
14 GARCIA MD, QUILEZ AM, SAENZ MT, MARTINEZ DOMINGUEZ ME, DE LA PUERTA R, 2000.
Anti-inflammatory activity of Agave intermixta Trel. and Cissus sicyoides L., species used in South Caribbean traditional medicine. J Ethnopharmacol 71(3):395-400.
15 MARTINEZ MJ, LOPEZ M, BOUCOURT E, BETANCOURT J, FUENTES V, MORON F, 2002.
Irritabilidad dérmica primaria de Cissus verticillata. Informe TRAMIL. Laboratorio Central de Farmacología, Facultad de Ciencias Médicas “Dr. Salvador Allende”, La Habana, Cuba.
16 MARTINEZ MJ, LOPEZ M, BOUCOURT E, BETANCOURT J, FUENTES V, MORON F, 2002.
Toxicidad aguda tópica de Cissus verticillata. Informe TRAMIL. Laboratorio Central de Farmacología, Facultad de Ciencias Médicas “Dr. Salvador Allende”, La Habana, Cuba.
17 CAMBAR P, 1992
Efecto de los extractos acuosos de Cissus verticillata L. Informe TRAMIL. Unidad de Farmacología, Facultad de Ciencias Médicas, Universidad Nacional de Honduras UNAH, Tegucigalpa, Honduras.