Cornutia pyramidata
Vernacular names
(In territories with significant traditional TRAMIL use)
Dominica:
- bwa kasav
Geographical distribution
Caribbean basin and northern South America.
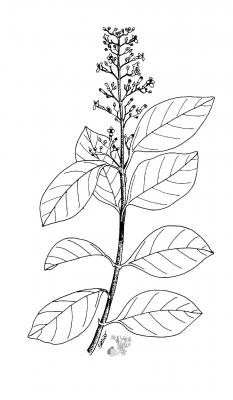
Botanical description
Tree or shrub 5 m or more; upright, quadrangular branches. Single, opposite, pubescent, ovate or elliptic leaves, 6 to 20 cm long, entire margin. Terminal pyramidal panicles, 10 to 40 cm long; numerous blue or purple flowers, 10 to 12 cm long. Blue or black drupe, spherical, 3 to 6 mm in diameter.
Voucher(s)
Zanoni, 44409, JBSD
Pharmacopoeia
Ed.3References
1 CHARLES C, 1988
TRAMIL survey. Movement for Cultural Awareness MCA, Roseau, Dominica.
2 PINEDA M, 1990
Fitoquímica de algunas plantas TRAMIL. Laboratorio de enda-caribe, Santo Domingo,Rep. Dominicana.
3 HERRERA J, 1990
Determinación de actividades biológicas de vegetales utilizados en medicina tradicional. Informe TRAMIL. Dep. de Farmacología, Facultad de Salud, Universidad del Valle, Cali, Colombia.