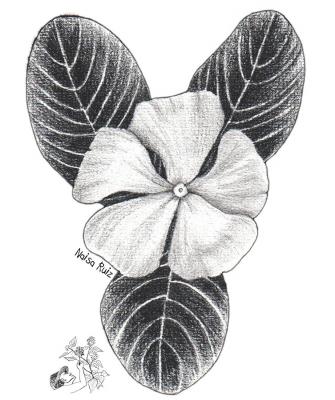
Catharanthus roseus
Vernacular names
(In territories with significant traditional TRAMIL use)
Honduras:
- guajaca
Cuba:
- vicaria
- vicaria blanca
Geographical distribution
Native to Madagascar, widely cultivated and naturalised in the tropical regions.
Botanical description
Herbaceous or subshrub of up to 80 cm, with milky white latex. Opposite obovate, elliptic or oblong leaves, 3 to 7 cm long. Sessile flowers, white or pink, with 5 petals joined by the base forming a tube with free lobes, solitary or in groups in the axils of the leaves. Trilobular fruit, pubescent, containing 2 to 3 black seeds, about 0.5 cm in diameter.
Voucher(s)
L.R.Soberats,TR90-06,CIFMT
Fuentes,4757,ROIG
Pharmacopoeia
Ed.3References
1 CARBALLO A, 1990
Encuesta TRAMIL. Centro de investigación de fitoterapia y medicina tradicional de Topes de Collantes, Trinidad, Cuba.
2 LAGOS-WITTE S, 1988-89, 1996
Encuesta TRAMIL. Laboratorio de Histología Vegetal y Etnobotánica, Dep. de Biología, Universidad Nacional Autónoma de Honduras UNAH, Tegucigalpa, Honduras.
3 MOREJON Z, LOPEZ M, GARCIA MJ, BOUCOURT E, VICTORIA M, FUENTES V, MORON F, BOULOGNE I, ROBINEAU L, 2009
Encuesta TRAMIL preliminar a grupos de vecinos en los municipios 10 de Octubre, Lisa, Marianao, Habana del Este (Cojímar) en la Ciudad de la Habana. Laboratorio Central de Farmacología, Universidad de Ciencias Médicas de La Habana, Ciudad de La Habana, Cuba.
4 DAVEY JE, VAN STADEN J, DE LEEUW GTN, 1981
Endogenous cytokinin levels and development of flower virescence in Catharanthus roseus infected with mycoplasmas. Physiol Plant Pathol 19:193-200.
5 ALI I, 1990
The alkaloids in flowers of Catharanthus roseus (L.) G. Don. Gomal Univ J Res 10(1):27-31.
6 ROJAS MCN, CUELLAR MCA, 1981
Comparative microbiological studies of the alkaloids of Catharanthus roseus and other related compounds. Rev Cubana Farm 15(2):131-138.
7 WILLAMAN JJ, LI HL, 1970
Alkaloid-bearing plants and their contained alkaloids, 1957-1968. Lloydia 33S(1):1-286.
8 ATTA-UR-RAHMAN, ALI I, BASHIR M, 1984
Isolation and structural studies on the alkaloids in flowers of Catharanthus roseus. J Nat Prod 47(3):554-555.
9 SEVESTRE-RIGOUZZO M, NEF-CAMPA C, GHESQUIERE A, CHRESTIN H, 1992-1993
Genetic diversity and alkaloid production in Catharanthus roseus, C. trichophyllus
and their hybrids. Euphytica 66(1/2):151-159.
10 MILO J, 1981
Flower color inheritance and shoot and ajmalicine yield components in successive developmental stages of pure lines and F-1 hybrids in Catharanthus roseus (L.) G. Don. Thesis-MS-Hebrew University.
11 DE PINHO PG, GONÇALVES RF, VALENTÃO P, PEREIRA DM, SEABRA RM, ANDRADE PB, SOTTOMAYOR M, 2009
Volatile composition of Catharanthus roseus (L.) G. Don using solid-phase microextraction and gas chromatography/mass spectrometry. J Pharm Biomed Anal 49(3):674-685.
12 LARA G, LASTRA H, 1991
Determinación de vinblastina en el follaje de Catharanthus roseus G. Don. Rev Cubana Farm 25(1):56-62.
13 GARCIA G, GARCIA R, 1987
Efectos sobre las estructuras oculares de la decocción de flores de vicaria blanca. Informe TRAMIL. Laboratorio de Control Biológico, Industria Médico-Farmacéutica, La Habana, Cuba.
14 NEOGI N, BHATIA M, 1956
Biological investigation of Vinca rosea. Indian J Pharmacy 18:73.
15 SRINIVASAN D, NATHAN S, SURESH T, LAKSHMANA PERUMALSAMY P, 2001
Antimicrobial activity of certain Indian medicinal plants used in folkloric medicine. J of Ethnopharmacol 74(3):217-220.
16 ROJAS M, CUELLAR M, 1981
Comparative microbiological studies of the alkaloids of Catharanthus roseus and other related compounds. Rev Cubana Farm 15(2):131-138.
17 CHAUHAN S, AGRAWAL S, MATHUR R, GUPTA RK, 1979
Phosphatase activity in testis and prostate of rats treated with embelin and Vinca rosea extract. Experientia 35(9):1183-1185.
18 WEE Y, GOPALAKRISHNAKONE P, CHAN A, 1988
Poisonous plants in Singapore - a colour chart for identification with symptoms and signs of poisoning. Toxicon 26(1):47.