Vernacular names
(In territories with significant traditional TRAMIL use)
Dominican Republic:
- llantén
Guadeloupe:
- miyé
- plantain
Martinique:
- miyé
- plantain
Trinidad and Tobago:
- miyé
- plantain
Dominica:
- planten
Haiti:
- planten
Botanical description
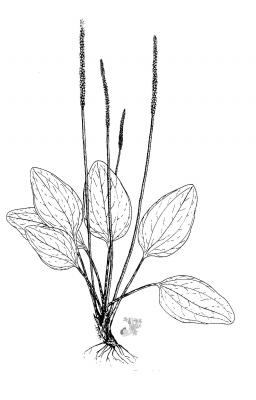
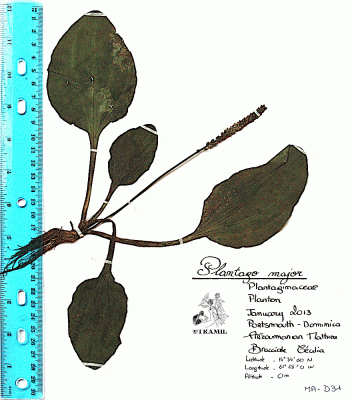
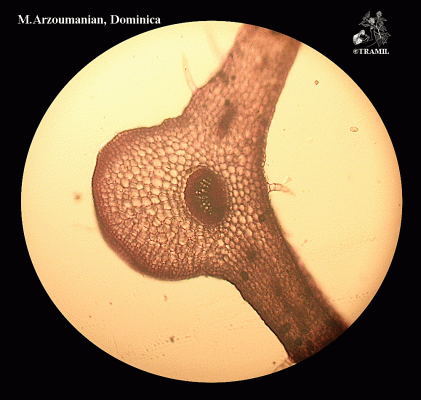
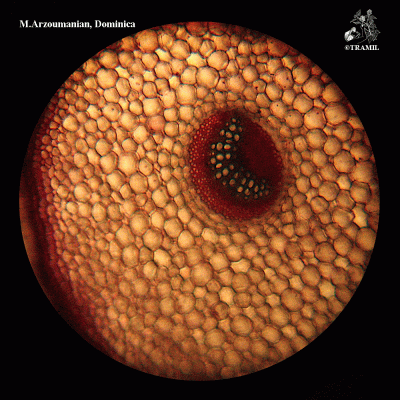
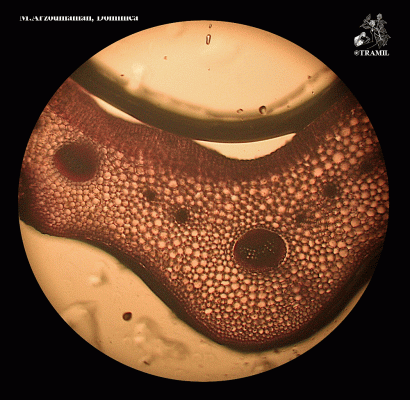
Annual or perennial acaulescent herb with a taproot producing numerous fibrous lateral roots. Leaves, glabrous, forming a basal rosette, petioles 2-15 cm long, blades ovate to elliptic, 2.5-25 x 3-12 cm, base abruptly narrow, apex obtuse, margins coarsely dentate; inflorescence a spike spike, linear-cylindrical, 5-25 cm x 6-8 mm, scape 10-30 cm. long; flowers perfect, corolla lobes 0.5-1 mm long, numerous, sessile, green; fruit a capsule ovate-elliptic 3-4 mm long, seeds brown, oblong 1 x 0.7 mm.
Voucher(s)
Rouzier,150,SOE
Fournet,5244,GUAD
Longuefosse&Nossin,29,HAVPMC
Delaigue,23,NHTT
Tórrez,6,CECALLI
Pharmacopoeia
Ed.2References
1 WENIGER B, ROUZIER M, 1986 Enquête TRAMIL. Service Oecuménique d'Entraide SOE, Port au Prince, Haïti.
2 CHARLES C, 1988 TRAMIL survey. Movement for Cultural Awareness MCA, Roseau, Dominica.
3 LONGUEFOSSE JL, NOSSIN E, 1990-95 Enquête TRAMIL. Association pour la valorisation des plantes médicinales de la Caraïbe AVPMC, Fort de France, Martinique.
4 GERMOSEN-ROBINEAU L, GERONIMO M, AMPARO C, 1984 Encuesta TRAMIL. enda-caribe, Santo Domingo, Rep. Dominicana.
5 EDOUARD JA, 1992 Enquête TRAMIL. Lycée agricole, Baie-Mahault, Guadeloupe.
6 LEBEDEV-KOSOV VI, 1980 Flavonoids and iridoids of Plantago major L. and Plantago asiatica L. Rast Resur 16:403-406.
7 MAKSYUTINA NP, 1971 Baicalein and scutellarein derivatives in Plantago major leaves. Khim Prir Soedin 7(3):374-375.
8 HARBORNE JB, WILLIAMS CA, 1971 Comparative biochemistry of flavonoids. XIII. 6-hydroxyluteolin and scutellarein as phyletic markers in higher plants. Phytochemistry 10:367-378.
9 ANDRZEJEWSKA-GOLEC E, SWIATEK L, 1984 Chemotaxonomic investigations on the genus plantago. I. Analysis of iridoid fraction. Herba Pol 30(1):9-16.
10 PAILER M, HASCHKE-HOFMEISTER E, 1969 Components of Plantago major. Planta Med 17(2):139-145.
11 MAKSYUTINA NP, 1971 Hydroxycinnamic acids from Plantago major and Plantago lanceolata. Khim Prir Soedin 7(6):824-825.
12 NORO Y, HISATA Y, OKUDA K, KAWAMURA T, KASAHARA Y, TANAKA T, SAKAI E, NISIBE S, SASAHARA M, 1991 Pharmacognostical studies of plantagins herba (VII) on the phenylethanoid contents ofPlantago spp. Shoyakugaku Zasshi 45(1):24-28.
13 JANSSON O, 1974 Hylloquinone (vitamin k-1) levels in leaves of plant species differing in susceptibility to 2,4-dichlorophenoxyacetic acid. Physiol Plant 31:323.
14 OBOLENTSEVA GV, KHADZHAI YI, 1966 Pharmacological testing of plantaglucide. Farmakol Toksikol 29(4):469-472.
15 BALBAA SI, KARAWYA MS, AFIFI MS, 1971 Pharmacognostical study of the seeds of certain plantago species growing in Egypt. U A R J Pharm Sci 12(1):35-52.
16 KUBOTA S, 1955 Chinese materia medica from which medicines have been introduced into Japan. Trans 9th Congr Far East Ass Trop Med 2:639.
17 ATAL CK, KAPOOR KK, SIDDIQUI HH, 1964 Studies on Indian seed oils. Part 1. Preliminary screening of linoleic acid rich oils. Indian J Pharmacy 26:163-164.
18 SAMUELSEN AB, COHEN EH, PAULSEN BS, BRULL LP, THOMAS-OATES JE, 1999 Structural studies of a heteroxylan from Plantago major L. seeds by partial hydrolysis, HPAEC-PAD, methylation and GC-MS, ESMS and ESMS/MS. Carbohydr Res 315(3/4):312-318.
19 DUKE JA, ATCHLEY AA, 1986 Handbook of proximate analysis tables of higher plants. Boca Raton, USA: CRC Press. p131.
20 SIDDIQUI M, HAKIM M, 1991 Crude drugs and their nutrient values. J of the National Integrated Med Assoc33(1):8-10.
21 CAMBAR P, 1989 Efecto antiinflamatorio del extracto acuoso de la hoja de llantén(Plantago major). Informe TRAMIL. Unidad de Farmacología, Facultad de Ciencias Médicas, Universidad Nacional Autónoma de Honduras UNAH, Tegucigalpa, Honduras. TRAMIL IV, Tela, Honduras, UNAH/enda-caribe.
22 CARBALLO A, 1995 Plantas medicinales del Escambray cubano. Informe TRAMIL. Laboratorio provincial de producción de medicamentos, Sancti Spiritus, Cuba.
23 PAZOS L, COTO T, GONZALEZ S, QUIROS S, 2004 Actividad sedante-tranquilizante en ratón, dosis repetidas del extracto acuoso de hoja de Plantago major. Informe TRAMIL.Laboratorio de Ensayos Biológicos, Universidad de Costa Rica, San Pedro, Costa Rica.
24 Lapa AJ, Souccar C, Lima-Landman MT, De Lima TC,2002 Métodos de evaluación de la actividad farmacológica de plantas medicinales. RIVAPLAMED. Red de validación de plantas medicinales. CYTED/CNPq. Programa Iberoamericano de Ciencia y Tecnología para el Desarrollo. Sao Paulo, Brasil.
25 CACERES A, GIRON LM, ALVARADO SR, TORRES MF, 1987 Screening of antimicrobial activity of plants popularly used in Guatemala for the treatment of dermatomucosal diseases. J Ethnopharmacol 20(3):223-237.
26 CAMBAR P, ALVARADO-GALVEZ C, ALGER J, RIVERA-VEGA O, 1984 Efectos broncopulmonares de algunas plantas medicinales de Honduras(conferencia). Unidad de Farmacología, Facultad de Ciencias Médicas, Universidad Nacional Autónoma de Honduras UNAH, Tegucigalpa, Honduras. II semana científica de la Universidad Nacional Autónoma de Honduras.
27 CAMBAR P, ALGER J, SANTOS A, 1983 Efectos farmacológicos de los extractos acuosos de las hojas de Llantén(Plantago major) (conferencia). Tegucigalpa, Honduras: XXVII Congreso médico de Honduras & I semana científica de la Universidad Nacional Autónoma de Honduras, Revista Médica Hondureña.
28 LAMBEV I, MARKOV M, PAVLOVA N, 1981 Study of the antiinflammatory and capillary restorative activity of a dispersed substance fromPlantago major L. Probl Nutr Med 9(3):162-169.
29 CAMBAR P, ALVARADO-GALVEZ C, ALGER J, RIVERA-VEGA O, 1984 Efectos broncopulmonares de algunas plantas medicinales de Honduras.(conferencia). Unidad de Farmacología, Facultad de Ciencias Médicas, Universidad Nacional Autónoma de Honduras UNAH, Tegucigalpa, Honduras. II semana científica de la Universidad Nacional Autónoma de Honduras.
30 CAMBAR P, SANTOS A, COUSIN L, 1985 Efecto del extracto acuoso de Plantago major (Llantén) en el choque anafiláctico inducido por albúmina de huevo en el conejo. Memoria de la III Semana Científica de la Universidad Nacional Autónoma de Honduras.
31 MIRONOV VA, VASIL'EV GS, MATROSOV VS, FILIPOVA TM, ZAMUREENKO VA,MISHCHENKO VV, MAIRANOVSKII VG, FEL'DSHTEIN MA,1983 Physiologically active alcohols from great plantain (Plantago major). Pharm Chem 17(11):794-798.
32 ANON, 1973 Traditional-western combined treatment of 217 cases of tetanus. Chung-hua I hsueh Tsa Chih Beijing53:682-684.
33 QUEIROZ I, REIS S, 1989 Antispasmodic and analgesic effects of some medicinal plants (conference). Rio, Brasil: Simpósio Brasil-China de Química e Farmacologia de Produtos Naturais, Abstr. Nº 180.
34 ROSA PINTO V, BARAHONA C, 1986 Estudio hipocrático de extractos acuosos de algunas plantas medicinales de uso tradicional en Honduras, sus efectos en ratas (Tesis de grado). Facultad de Química y Farmacia, Universidad Nacional Autónoma de Honduras, Tegucigalpa, Honduras.
35 DUCKETT S, 1980 Plantain leaf for poison ivy. N Engl J Med 303(10):583.
36 WAGNER H, 1987 Immunostimulants from higher plants. In HOSTETTMANN K, LEA PJ, (Eds.). Biologically active natural products.Oxford, UK: Oxford Science Publications, p127-141.
37 BETANCOURT J, MARTINEZ MJ, LOPEZ M, MOREJON Z, BOUCOURT E, MORON F, 2000 Actividad genotóxica in vitro de partes aéreas de Plantago major L. Informe TRAMIL. Laboratorio Central de Farmacología, Facultad de Ciencias. Médicas “Dr. Salvador. Allende”, La Habana, Cuba.
38 BETANCOURT J, MARTINEZ MJ, LOPEZ M, MOREJON Z, BARCELO H, LAINEZ A, MONTES ME, REGO R, BOUCOURT E, MORON F, 2000 Toxicidad aguda clásica de partes aéreas de Plantago major L. Informe TRAMIL. Laboratorio Central de Farmacología, Facultad de Ciencias. Médicas “Dr. Salvador. Allende”, La Habana, Cuba.
39 GarcIa GM, Coto MT, GonzAlez CS, Pazos L, 1996 Toxicidad por vía oral en ratón, del extracto acuoso de hojas frescas de Plantago major. Informe TRAMIL. Laboratorio de Ensayos Biológicos LEBI, Universidad de Costa Rica, San Pedro, Costa Rica.
40 GarcIa GM, Coto MT, GonzAlez CS, Pazos L, 1996 Irritabilidad ocular en conejos, del extracto acuoso de hojas frescas de Plantago major. Informe TRAMIL. Laboratorio de Ensayos Biológicos LEBI, Universidad de Costa Rica, San Pedro, Costa Rica.
41 ARROYO J, RODRIGUEZ N, RODRIGUEZ M, 1963 Cytostatic agents of plant and synthetic origin. Anales Real Acad Farm 29(4):157-169.
42 ALBORNOZ A, 1993 Medicina tradicional herbaria. Caracas, Venezuela: Instituto Farmacoterápico Latino S.A. p273.
43 DELAIGUE J, 2005 TRAMIL survey. UAG & PRDI, Tobago House of Assembly, Scarborough, Tobago.
44 GUERRA MJ, LOPEZ M, BOUCOURT E, FUENTES V, 2002 Irritabilidad oftálmica de la decocción 30% de hojas secas de Plantago major L.Informe TRAMIL. Laboratorio Central de Farmacología. Facultad de Medicina Dr. Salvador Allende. Ciudad de La Habana, Cuba.
45 LOGARTO PARRA A, SILVA YHEBRA R, GUERRA SARDINAS I, IGLESIAS BUELA L, 2001 Comparative study of the assay of Artemia salina L. and the estimate of the medium lethal dose (LD50 value) in mice, to determine oral acute toxicity of plant extracts. Phytomedicine 8(5):395-400.
46 LUCIANO-MONTALVO C, GAVILLAN-SUAREZ J, 2009 Actividades antimicrobianas de partes de plantas con usos significativos en encuestas etnofarmacológicas TRAMIL.Informe TRAMIL,Instituto de Investigaciones Interdisciplinarias, Cayey, Universidad de Puerto Rico.