Vernacular names
(In territories with significant traditional TRAMIL use)
Dominican Republic:
- bay run, berrón
Dominica:
- bay-run
- bwaden
Geographical distribution
Native to northern South America and the Antilles, cultivated in the tropics.
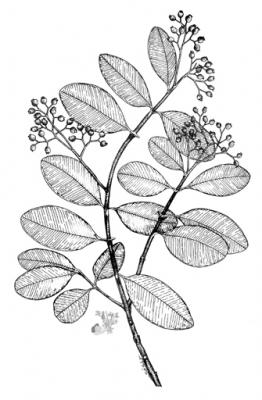
Botanical description
Tree of up to 15 m in height with grey to light brown bark that breaks off into thin sheets. Leaves elliptic, oblong or obovate,
obtuse at the base and emarginate at the apex, glabrous with finely reticulate venation, glandular-punctate. Corymbose, terminal or subterminal panicles; white or pink, glandular flowers with numerous stamens; calyx with 5 lobes, broadly triangular. Ovoid fruit of up to 1 cm, black when ripe.
Voucher(s)
Jiménez,60,JBSD
Pharmacopoeia
Ed.3References
1 GERMOSEN-ROBINEAU L, GERONIMO M, AMPARO C, 1984
Encuesta TRAMIL. enda-caribe, Santo Domingo, Rep. Dominicana.
2 CHARLES C, 1988
TRAMIL survey. Movement for Cultural Awareness MCA, Roseau, Dominica.
3 BOURGEOIS P, 1986
Rapport concernant Pimenta racemosa (Myrtacées). Rapport TRAMIL. Laboratoire de phytochimie, Faculté des Sciences, UAG, Guadeloupe.
4 FURIA T, BELLANCA N, 1971
Fenaroli’s handbook of flavour ingredients. Cleveland, USA: The Chemical Rubber Co.
5 GARCIA D, ALVAREZ A, TORNOS P, FERNANDEZ A, SÁENZ T, 2002
Gas chromatographic-mass spectrometry study of the essential oils of Pimenta racemosa var. terebinthina and P.racemosa var grisea. Z Naturforsch C 57(5-6):449-451.
6 LEYVA M, TARACONTE J, MARQUETTI MdC, 2007
Composición química y efecto letal del aceite esencial de Pimenta racemosa (Myrtales: Myrtaceae) sobre Blattella
germanica (Dictyoptera: Blattellidae). Rev Cubana Med Trop 59(2):154-158.
7 LEUNG AY, FOSTER S, 1980
Encyclopedia of common natural ingredients used in food, drugs and cosmetics. New York, USA: Wiley Interscience.
8 NEGWER M, 1987
Organic chemical drugs and their synonyms (an international survey), 6th ed. Berlin, Germany: Akademie Verlag.
9 DUKE JA, 1992
Handbook of biologically active phytochemicals and their bioactivities. Boca Raton, USA: CRC Press.
10 DE SOUSA M, MATOS ME, MATOS FJ, MACHADO MI, CRAVEIRO AA, 1991
Constituintes químicos ativos de plantas medicinais Brasileiras. Fortaleza, Brasil: Ceará Edições UFC Laboratorio de produtos naturais.
11 MAC GREGOR JT, LAYTON LL, BUTTERY RG, 1974
California bay oil. II. Biological effects of constituents. J Agric Food Chem 22(5):777-780.
12 UNGSURUNGSIE M, SUTHIENKUL O, PAOVALO C, 1982
Mutagenicity screening of popular Thai spices. Food Chem Toxicol 20(5):527-530.
13 HERRERA J, 1988
Determinación de actividades biológicas de vegetales utilizados en medicina tradicional. Informe TRAMIL. Dep. de Farmacología, Facultad de Salud, Universidad del Valle, Cali, Colombia.
14 BUDAVARI S (Ed.), 2001
The Merck Index: an encyclopedia of chemicals, drugs, and biologicals. 30th ed. Whitehouse Station, USA: Merck & Co., Inc. p690.
15 REYNOLDS J (Ed.), 1996
Martindale: The extra pharmacopoeia. 31st ed. London, England: The Royal Pharmaceutical Society. p1705.
16 CARBALLO A, 1995
Cálculo de concentración y dosis de las drogas vegetales TRAMIL: Mensuraciones farmacognósticas y aproximaciones técnico-clínicas. Laboratorio provincial de producción de medicamentos, Sancti Spiritus, Cuba.