Vernacular names
(In territories with significant traditional TRAMIL use)
Guatemala:
- aguacate
Dominican Republic:
- aguacate
Mexico:
- aguacate
Barbados:
- pear tree
Martinique:
- zaboka
Botanical description
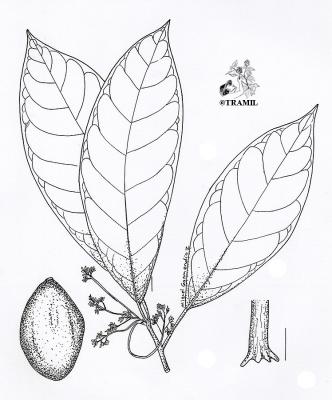
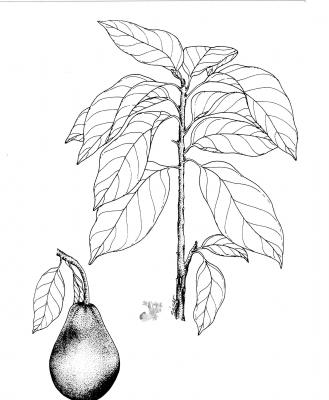
Tree 14-40 m high. Leaves alternate, simple, entire, elliptic to obovate, acute or acuminate at the tip, cuneate or rounded at the base, penninerved, 6-30 cm x 3.5-19 cm. upper surface glabrescent, lower surface glaucose; inflorescence axilary, compact or loose panicles; flowers perfect, greenish-yellowish, perianth segments 4-6 mm long; fruit a pear-shaped drupe, shiny, green or purplish, up to ca.15 cm long; seed fleshy, loose in the pericarp at maturity.
Voucher(s)
Girón,245,CFEH
Longuefosse&Nossin,25,HAVPM
Medina,60,CICY
Pharmacopoeia
Ed.2References
1 GERMOSEN-ROBINEAU L, GERONIMO M, AMPARO C, 1984 Encuesta TRAMIL. enda-caribe, Santo Domingo, Rep. Dominicana.
2 GIRON L, 1988 Encuesta TRAMIL (Costa atlántica). Centro Mesoamericano de Tecnología CEMAT, Guatemala, Guatemala.
3 LONGUEFOSSE JL, NOSSIN E, 1990-95 Enquête TRAMIL. Association pour la valorisation des plantes médicinales de la Caraïbe AVPMC, Fort de France, Martinique.
4 MENDEZ M, MEDINA ML, DURAN R, 1996 Encuesta TRAMIL. Unidad de recursos naturales, Centro de Investigación Científica de Yucatán CICY, Mérida, México.
5 CANIGUERAL S, VILA R, RISCO E, PEREZ F, PORTILLO A, FREIXA B, MILO B, VANACLOCHA B, RIOS JL, MORALES MA, ALONSO JR, BACHILLER LI, PERIS JB, STUBING G, 2002 Persea americana. Vademecum de Fitoterapia, Editorial Masson, Barcelona, España, Feb. 26, 2003. URL: http://www.masson.es/book/fitoterapia.html
6 BERGH BO, SCORA RW, STOREY WB, 1973 Comparison of leaf terpenes in Persea subgenus persea. Bot Gaz (Chicago) 134:130-134.
7 KING JR, KNIGHT RJ, 1992 Volatile components of the leaves of various avocado cultivars. J Agric Food Chem 40(7):1182-1185.
8 DE ALMEIDA AP, MIRANDA MMFS, SIMONI IC, WIGG MD, LAGROTA MHC, COSTA SS, 1998 Flavonol monoglycosides isolated from the antiviral fractions of Persea americana (Lauraceae) leaf infusion. Phytother Res 12(8):562-567.
9 MERIÇLI F, MERIÇLI AH, YILMAZ F, YÜNCÜLER G, YÜNCÜLER O, 1992 Flavonoids of avocado (Persea americana) leaves. Acta Pharm Turc 34(2):61-63.
10 BATE-SMITH EC, 1975 Phytochemistry of proanthocyanidins. Phytochemistry 14(4):1107-1113.
11 MURAKOSHI S, ISOGAI A, CHANG CF, KAMIKADO T, SAKURAI A, TAMURA S, 1976 The effects of two components from avocado leaves (Persea americana) and related compounds on the growth of silkworm larvae, Bombyx mori. Nippon Oyo Dobutsu Konchu Gakkaishi 20:87-91.
12 HIRAI N, KOSHIMIZU K, 1983 A new conjugate of dihydrophaseic acid from avocado fruit. Agr Biol Chem 47(2):365-371.
13 WILSON C, WILSON III CW, SAW PE, NAGY S, 1979 Analysis of monosaccharides in avocado by HPLC. Liq Chromat Anal Food Beverages 1:225-236.
14 SARDI JC, TORRES OA, 1978 Study on avocado (Persea americana) oil. Arch Bioquim Quim Farm 20:45-49.
15 DUKE JA, ATCHLEY AA, 1986 Handbook of proximate analysis tables of higher plants. Boca Raton, USA: CRC Press. p324.
16 CACERES A, GONZALEZ S, GIRON L, 1998 Demostración de la actividad antimicrobiana de plantas TRAMIL en base a los usos populares en la cuenca del Caribe. Informe TRAMIL. Laboratorio de productos fitofarmacéuticos Farmaya y Facultad de ciencias químicas y farmacia, Universidad de San Carlos, Guatemala, Guatemala.
17 HERRERA J, 1986 Determinación de actividades biológicas de vegetales utilizados en medicina tradicional. Informe TRAMIL. Dep. de Farmacología, Facultad de Salud, Universidad del Valle, Cali, Colombia.
18 GarcIa GM, Coto MT, GonzAlez CS, Pazos L, 1999 Actividad bronquial del extracto acuoso de hoja fresca de Persea americana. Informe TRAMIL. Laboratorio de Ensayos Biológicos LEBI, Escuela de Medicina, Universidad de Costa Rica, San Pedro, Costa Rica.
19 PAZOS L, COTO T, GONZALEZ S, QUIROS S, 2003 Tránsito intestinal, en ratones, del extracto acuoso de hojas frescas de Persea americana. Informe TRAMIL. Laboratorio de Ensayos Biológicos, Universidad de Costa Rica, San Pedro, Costa Rica.
20 ADEYEMI OO, OKPO SO, OGUNTI OO, 2002 Analgesic and anti-inflammatory effects of the aqueous extract of leaves of Persea americana Mill Lauraceae. Fitoterapia 73(5):375-380.
21 MIWA M, KONG ZL, SHINOHARA K, WATANABE M, 1990 Macrophage stimulating activity of foods. Agric Biol Chem 54(7):1863-1866.
22 HERRERA J, 1988 Determinación de actividades biológicas de vegetales utilizados en medicina tradicional. Informe TRAMIL. Laboratorio de fitofarmacología, Dep. de Farmacología, Facultad de Salud, Universidad del Valle, Cali, Colombia.
23 GarcIa GM, Coto MT, GonzAlez CS, Pazos L, 2000 Toxicidad aguda en ratones, del extracto acuoso de hojas frescas de Persea americana. Informe TRAMIL. Laboratorio de Ensayos Biológicos LEBI, Escuela de Medicina, Universidad de Costa Rica, San Pedro, Costa Rica.
24 CRAIGMILL AL, SEAWRIGHT AA, MATTILA T, FROST AJ, 1989 Pathological changes in the mammary gland and biochemical changes in milk of the goat following oral dosing with leaf of the avocado (Persea americana). Aust Vet J 66(7):206-211.
25 GRANT R, BASSON PA, BOOKER HH, HOFHERR JB, ANTHONISSEN M, 1991 Cardiomyopathy caused by avocado (Persea americana Mill.) leaves. J S Afr Vet Assoc 62(1):21-22.
26 ALONSO J, 1998 Tratado de fitomedicina. Bases clínicas y farmacológicas. Buenos Aires, Argentina: ISIS ediciones SRL. p185.
27 LOPEZ M, MARTINEZ MJ, MOREJON Z, BOUCOURT E, FUENTES V, MORON F. 2005 Irritabilidad dérmica primaria de hoja fresca machacada de Persea americana Mill. Informe TRAMIL. Laboratorio Central de Farmacología, Facultad de Medicina “Dr. Salvador Allende”, Cerro, C. Habana, Cuba.
28 MORON FJ, GARCIA AI, VICTORIA MC, MOREJON Z, LOPEZ M, BACALLAO Y, FUENTES V, 2008 Acción analgésica de la decocción de hojas frescas de Persea americana Mill. (aguacate) en ratones. Trabajo TRAMIL. Laboratorio Central de Farmacología. Universidad de Ciencias Médicas de La Habana.