Vernacular names
(In territories with significant traditional TRAMIL use)
Martinique:
- mannyòk
- manyok
Haiti:
- mannyòk
- manyok
Dominican Republic:
- yuca
Botanical description
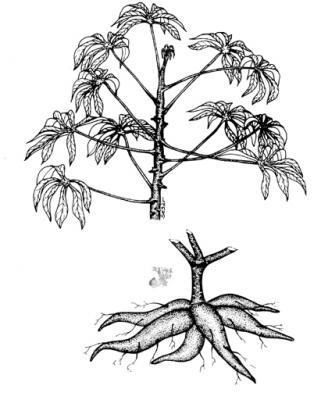
Herb or semiwoody monecousshrub, 1-3 m high, with large, tuberous roots. Leaves alternate, spirally arranged, palmately 3-9 lobed, obovate-lanceolate, 4-20 cm x 1-6 cm, glabrous above and glaucous below, petiole 5-30 cm; inflorescence terminal cymose panicles; female flowers on pedicels 7-12 mm long, calyx lobe 7.5 – 10 mm long, male flowers on pedicels 4-6 mm long, calyx lobes 2-5 mm long, female flowers lower than the male flowers; fruit a dehiscent, globose capsule, 1.5 cm in long; seeds gray mottled,ellipsoid, ca.12 mm long.
Voucher(s)
Rouzier,63,SOE
Jiménez,1523,JBSD
Pharmacopoeia
Ed.2References
1 WENIGER B, ROUZIER M, 1986 Enquête TRAMIL. Service Oecuménique d'Entraide SOE, Port au Prince, Haïti.
2 LONGUEFOSSE JL, NOSSIN E, 1990-95 Enquête TRAMIL. Association pour la valorisation des plantes médicinales de la Caraïbe AVPMC, Fort de France, Martinique.
3 WHO, 1991 Guidelines for the assessment of herbal medicines. WHO/TRM/91.4. Programme on Traditional Medicines, WHO, Geneva, Switzerland.
4 LINDNER E, 1995 Toxicología de los alimentos. 4ª ed. Madrid, España: Editorial Acribia S.A. p688.
5 ROSA DE BATTISTI C, TELES F, COELHO D, JOSE DA SILVEIRA A, BATISTA C, 1981 Determination of hydrogen cyanide toxicity and total soluble carbohydrates in cassava (Manihot esculenta Crantz). Rev Ceres 28:521-525.
6 VALYASEVI A, DHANAMITTA S, 1974 Studies of bladder stone disease in Thailand. XVII. Effect of exogenous sources of oxalate on crystalluria. Amer J Clin Nutr 27(8):877-882.
7 KAMIL M, ILYAS M, RAHMAN W, OKIGAWA M, KAWANO N, 1994 Biflavones from Manihot utilissima. Phytochemistry 13(11):2619-2620.
8 SUBRAMANIAN S, NAGARAJAN S, SULOCHANA N, 1971 Flavonoids of some Euphorbiaceous plants. Phytochemistry 10:2548-2549.
9 SAKAI T, NAKAGAWA Y, 1988 Diterpenic stress metabolites from cassava roots. Phytochemistry 27(12):3769-3779.
10 LA LAGUNA F, 1993 Purification of fresh cassava root polyphenols by solid-phase extraction with Amberlite xad-8 resin. J Chromatogr A 657(2):445-449.
11 LYKKESFELDT J, MOLLER BL, 1994 Cyanogenic glucosides in cassava, Manihot esculenta Crantz. Acta Chem Scand 48(2):178-180.
12 DUKE JA, ATCHLEY AA, 1986 Handbook of proximate analysis tables of higher plants. Boca Raton, USA: CRC Press.
13 CACERES A, 2000 Actividad antibiótica in vitro del zumo de hoja fresca deManihot esculenta. Informe TRAMIL. Facultad de Ciencias Químicas y Farmacia, Universidad de San Carlos USAC, Guatemala, Guatemala.
14 MACRAE WD, HUDSON JB, TOWERS GH, 1988 Studies on the pharmacological activity of Amazonian Euphorbiaceae. J Ethnopharmacol 22(2):143-172.
15 GASPERI-CAMPANI A, BARBIERI L, BATTELLI MG, STIRPE F, 1985 On the distribution of ribosome-inactivating proteins amongst plants. J Nat Prod 48(3):446-454.
16 ITOKAWA H, HIRAYAMA F, TSURUOKA S, MIZUNO K, TAKEYA K, NITTA A, 1990 Screening test for antitumor activity of crude drugs (III). Studies on antitumor activity of Indonesian medicinal plants. Shoyakugaku Zasshi 44(1):58-62.
17 FERNANDO R, 1988 Plant poisoning in Sri Lanka. Toxicon 26(1):20.
18 ALONSO J, 1998 Tratado de fitomedicina. Bases clínicas y farmacológicas. Buenos Aires, Argentina: ISIS ediciones SRL. p687.
19 MADUAGWU EN, UMOH IB, 1982 Detoxification of cassava leaves by simple traditional methods. Toxicol Lett 10(2-3):245-248.
20 LOPEZ M, MARTINEZ MJ, MOREJON Z, BOUCOURT E, FUENTES V, MORON F. 2005 Irritabilidad dérmica primaria de tubérculo fresco rayado de Manihot esculenta Crantz. Informe TRAMIL. Laboratorio Central de Farmacología, Facultad de Medicina “Dr. Salvador Allende”, Cerro, C. Habana, Cuba.
21 PAZOS L, COTO T, GONZALEZ S, 2006 Irritabilidad dérmica, piel sana en conejos, de hoja machacada de Manihot esculenta. Informe TRAMIL. Laboratorio de Ensayos Biológicos, LEBi, Universidad de Costa Rica, San Pedro, Costa Rica.