Vernacular names
(In territories with significant traditional TRAMIL use)
Dominican Republic:
- mamey
Botanical description
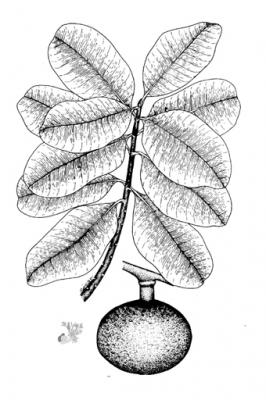
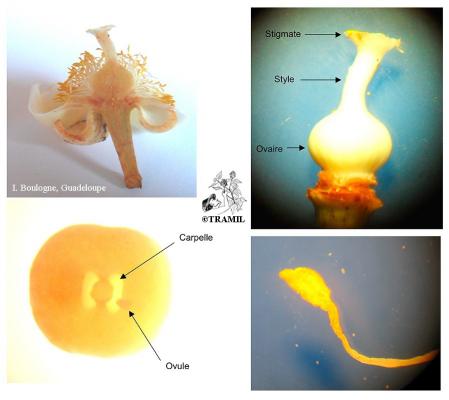
Tree, up to 20 m high. Leaves opposite, 8-25 cm x 10 cm, elliptic to obovate, glossy, dark green with pellucid dots, exuding a milky sap when broken; Flowers axillary, solitary or few together, fragrant, with white obovate petals ca. 20 mm long; fruit a globose drupe ca. 15 cm in diameter, skin rough, brown and thick; seeds 1-4 fibrous coated stone embedded in sweet, yellow pulp.
Voucher(s)
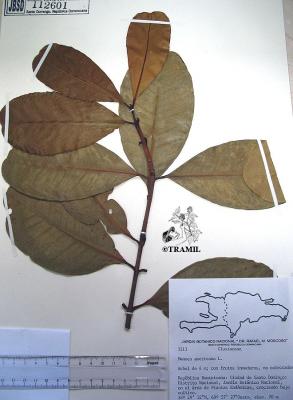
Jiménez,698,JBSD
Veloz,3113,JBSD
Pharmacopoeia
Ed.2References
1 GERMOSEN-ROBINEAU L, GERONIMO M, AMPARO C, 1984 Encuesta TRAMIL. enda-caribe, Santo Domingo, Rep. Dominicana.
2 PLOUVIER V, 1964 L-inositol, L-quebrachitol, and D-pinitol in some botanical groups. The presence of shikimic acid in Mammea americana. CR ACAD SCI 258(10):2921-2924.
3 SASAK W, CHOJNACKI T, 1973 Long chain polyphenols of tropical and subtropical plants. Acta Biochim Pol 20:343-350.
4 GAMES DE, 1972 Identification of 4-phenyl and 4-alkylcoumarins inMammea americana L.,Mammea africana G. Don. andCalophyllum inophyllum by gas chromatography-mass spectrometry. Tetrahedron Lett 31:3187-3190.
5 FINNEGAN R, PATEL J, BACHMAN P, 1966 Constituents ofMammea americana L. V (1). Some simple mono- and di-hydroxyxanthones. Tetrahedron Lett 49:6087-6092.
6 FINNEGAN R, MERKEL K, BACK N, 1972 Constituents ofMammea americana L. VIII: Novel structural variations on the mammein theme and antitumor activity of mammein and related coumarin and phloroglucinol derivatives. J Pharm Sci 61(10):1599-1603.
7 FINNEGAN R, MUELLER W, 1965 Constituents ofMammea americana L. IV. The structure of mammeigin. J Org Chem 30(7):2342-2344.
8 CROMBIE L, GAMES D, HASKINS N, REED G, 1972 Extractives ofMammea americana L. Part IV. Identification of new 7,8-annulated relatives of the coumarinsMammea A/AA, A/AB, B/AA B/AB, and new members of the 6-acyl family A/BB, B/AB, and B/AC. J Chem Soc Perkin Trans 18:2248-2254.
9 FINNEGAN RA, MERKEL KE, 1972 Constituents ofMammea americana L. IX. Oxidation of mammein and mammeisin. J Pharm Sci 61(10):1603-1608.
10 FINNEGAN R, PATEL J, 1972 Constituents ofMammea americana L. Part X. The isolation of some mono- and di-hydroxyxanthones. Observations on the synthesis of 1,5-; 3,5-; 1,6-; and 1,7-dihydroxyxanthone. J Chem Soc Perkin Trans I 15:1896-1901.
11 CROMBIE L, GAMES D, Mc CORMICK A, 1967 Extractives ofMammea americana L. I. The 4-N-alkylcoumarins. Isolation & structure ofMammea B/BA, B/BB, B/BC and C/BB. J Chem Soc C 23:2545-2552.
12 DJERASSI C, EISENBRAUN E, FINNEGAN R, GILBERT B, 1960 Naturally occurring oxygen heterocyclics. VII. The structure of mammein. J Org Chem 25:2164-2169.
13 PAZOS L, COTO T, GONZALEZ S, 2006 Antiinflamatorio, tópico, en ratón, de la decocción de hoja de Mammea americana. Informe TRAMIL. Laboratorio de Ensayos BiológicosLEBi, Escuela de MedicinaUniversidad de Costa Rica, San Pedro, Costa Rica.
14 SIEVERS AF, ARCHER WA, MOORE RH, MC GOVRAN BR, 1949 Insecticidal tests of plants from tropical America. J Econ Entomol 42(3):549-551.
15 PLANK HK, 1944 Insecticidal properties of mamey and other plants in Puerto Rico. J Econ Entomol 37(6):737-739.
16 FINNEGAN RA, MERKEL KE, PATEL JK, 1973 Constituents ofMammea americana L. XII. Biological data for xanthones & benzophenones. J Pharm Sci 62(3):483-485.
17 GRAINGE M, AHMED S, 1988 Handbook of plants with pest-control properties. New York, USA: John Willey & Sons. p178.
18 SUKH D, OPENDER K, 1997 Insecticides of natural origin. Amsterdam, Holland: Overseas Publishers Association. p197.
19 GarcIa GM, Coto MT, GonzAlez S, OCAMPO R, Pazos L, 2000 Irritabilidad dérmica del extracto acuoso de hoja fresca de Mammea americana. Informe TRAMIL. Laboratorio de Ensayos Biológicos LEBi, Escuela de Medicina, Universidad de Costa Rica, San Pedro, Costa Rica.