Vernacular names
(In territories with significant traditional TRAMIL use)
Barbados:
- banana
Haiti:
- bannan matenten
Dominican Republic:
- rulo
Botanical description
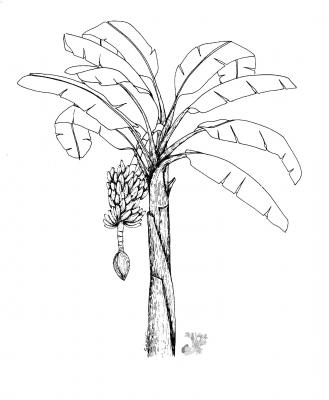
Large perennial herb 2-9 m high with basal corm. Pseudostem composed of leaf sheaths forming the ‘trunk’ of the plant. Leaves blades oblong, entire 1.5-4 m x 0.7-1 m, petiole 30-90 cm long, rounded beneath, channeled above; inflorescences terminal, growing from the pseudostem, erect or drooping; flowers in flat clusters in the axils of spirally arranged broad, dark purple-coloured bracts, functionally unisexual, the lower female the upper male; mature fruit fleshy, cylindrical, 6-35 cm x 2.5-5 cm with greenish, yellow or reddish skin.
Voucher(s)
Faujour,7,BAR
Jiménez,691,JBSD
Longuefosse&Nossin,24,HAVPM
Pharmacopoeia
Ed.2References
1 SIMMONDS NW, 1962 The evolution of the bananas. London, England: Longmans, Green & Co. Ltd. p170.
2 WENIGER B, ROUZIER M, 1986 Enquête TRAMIL. Service Oecuménique d'Entraide SOE, Port au Prince, Haïti.
3 FAUJOUR A, MURREY D, CHELTENHAM-CORBIN B, CARRINGTON S, 2003 TRAMIL survey. enda-caribbean, IICA & UAG, Saint Thomas, Barbados.
4 GERMOSEN-ROBINEAU L, GERONIMO M, AMPARO C, 1984 Encuesta TRAMIL. enda-caribe, Santo Domingo, Rep. Dominicana.
5 WHO, 1991 Guidelines for the assessment of herbal medicines. WHO/TRM/91.4. Programme on Traditional Medicines, WHO, Geneva, Switzerland.
6 PALMER J, WYMAN H, 1965 Organic acids in banana leaves. Phytochemistry 4(2):305-309.
7 WONG W, 1976 Some folk medicinal plants from Trinidad. Econ Bot 30:103-142.
8 DUTTA PK, DAS AK, BANERJI N, 1983 A tetracyclic triterpenoid from Musa paradisiaca. Phytochemistry22(11):2563-2564.
9 WILLAMAN JJ, HUI-LIN L, 1970 Alkaloid-bearing plants and their contained alkaloids, 1957-1968. Lloydia33(3A)Supp.
10 FRIESE FW, 1934 Plantas medicinais brasileiras. Sao Paulo, Brasil: Inst Agro do Estado Sao Paulo. p252-494.
11 DUKE JA, ATCHLEY AA, 1986 Handbook of proximate analysis tables of higher plants.Boca Raton, USA: CRC Press.
12 RICHTER E, VORE L, 1989 Antimicrobial activity of banana puree. Food Microbiol 6(3):179-187.
13 ALONSO J, 1998 Tratado de fitomedicina: bases clínicas y farmacológicas. Buenos Aires, Argentina: ISIS ediciones SRL. p125-126.
14 NEGWER M, 1987 Organic chemical drugs and their synonyms (an international survey). 6th ed. Berlin, Germany: Akademie Verlag.
15 CARBALLO A, 1995 Plantas medicinales del Escambray cubano. Informe TRAMIL. Laboratorio provincial de producción de medicamentos, Sancti Spiritus, Cuba.
16 CARBALLO A, 1995 Cálculo de concentración y dosis de las drogas vegetales TRAMIL: Mensuraciones farmacognósticas y aproximaciones técnico-clínicas. Laboratorio provincial de producción de medicamentos, Sancti Spiritus, Cuba.
17 PAZOS L, COTO T, CAIZA F, 2009
Irritación dérmica, piel lesionada en conejos, de la savia del fruto verde de Musa paradisiaca. Informe TRAMIL. Laboratorio de Ensayos Biológicos, LEBi, Universidad de Costa Rica, San Pedro, Costa Rica.
18 PAZOS L, COTO T, CAIZA F, 2009
Irritación dérmica, piel lesionada en conejos, del mesodermo del fruto maduro de Musa paradisiaca. Informe TRAMIL. Laboratorio de Ensayos Biológicos, LEBi, Universidad de Costa Rica, San Pedro, Costa Rica.
19 PAZOS L, COTO T, CAIZA F, 2009
Irritación dérmica, piel sana en conejos, porción de hoja calentada de Musa paradisiaca. Informe TRAMIL. Laboratorio de Ensayos Biológicos, LEBi, Universidad de Costa Rica, San Pedro, Costa Rica.
20 MORON FJ, GARCIA AI, VICTORIA MC, MOREJON Z, LOPEZ M, BACALLAO Y, FUENTES V, 2008 Acción analgésica oral de la decocción de hojas frescas de Musa x paradisiaca L. (plátano) en ratones. Trabajo TRAMIL. Laboratorio Central de Farmacología. Universidad de Ciencias Médicas de La Habana.
21 FRIAS AI, GARCIA N, MOREJON Z, MORON F, VICTORIA MC, 2009 Efecto antiinflamatorio tópico de la decocción de hojas frescas de Musa x paradisiaca L. (plátano) en el edema de la oreja inducido por aceite de Croton en ratones. Trabajo TRAMIL. Laboratorio Central de Farmacología. Universidad de Ciencias Médicas de La Habana.
22 MORON FJ, MOREJON Z, VICTORIA MC, BACALLAO Y, FUENTES V, 2009 Acción sobre el tránsito intestinal del zumo fresco del fruto maduro de Musa x paradisiaca L. (plátano) en ratones. Trabajo TRAMIL. Laboratorio Central de Farmacología. Universidad de Ciencias Médicas de La Habana.