Vernacular names
(In territories with significant traditional TRAMIL use)
Martinique:
- chadwon beni
- fit weed
Dominica:
- chadwon beni
- fit weed
Trinidad and Tobago:
- chadwon beni
- fit weed
Dominican Republic:
- cilantro ancho
- koulant
Honduras:
- culantro
Costa Rica:
- culantro de coyote
- spirit weed
Haiti:
- koulant
Geographical distribution
Native to tropical America, introduced and naturalised in tropical Africa.
Botanical description
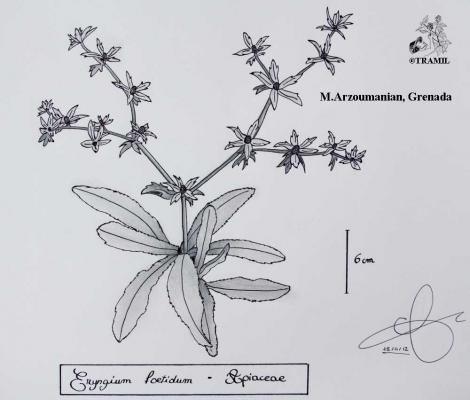
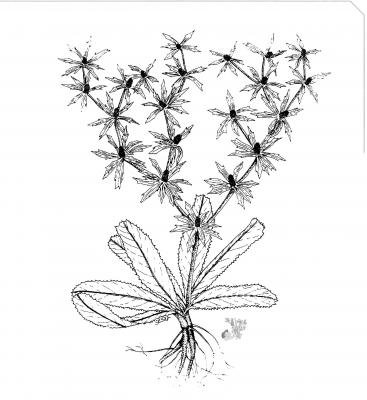
Perennial herb, glabrous, up to 60 cm Lanceolate to oblanceolate leaves up to 30 cm long, crenate to finely spiny serrated, thinned at the base forming a short, winged petiole. Inflorescences in dichasia, extensively ramified with green- yellowish capitula; involucral bracts, lanceolate leaflets, extending beyond the capitula. Globose fruit, laterally compressed, densely scaly.
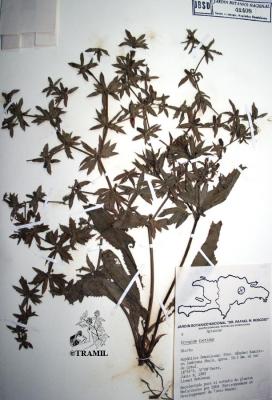
Voucher(s)
Ocampo,5013,CR
Ochoa,280,HPMHV
Jiménez,125,JBSD Jean Pierre,105,SLNH Delaigue,12,NHTT
Longuefosse&Nossin,16,HAVPM
Pharmacopoeia
Ed.3References
- OCAMPO R, 1988
Encuesta TRAMIL (Costa atlántica), Instituto de Desarrollo Agrario, Universidad de Costa Rica, San José, Costa Rica.
- CHARLES C, 1988
TRAMIL survey. Movement for Cultural Awareness MCA, Roseau, Dominica.
- JEAN-PIERRE L, 1988
TRAMIL survey. St. Lucia National Herbarium, Castries, St. Lucia.
- LONGUEFOSSE JL, NOSSIN E, 1990-95
Enquête TRAMIL. Association pour la valorisation des plantes médicinales de la Caraïbe AVPMC, Fort de France, Martinique.
- DELAIGUE J, 2005
TRAMIL survey. PRDI, Tobago House of Assembly, Scarborough, Tobago.
- GERMOSEN-ROBINEAU L, GERONIMO M, AMPARO C, 1984
Encuesta TRAMIL. enda-caribe, Santo Domingo, Rep. Dominicana.
- LAGOS-WITTE S, 1988-1995
Encuestas TRAMIL. Laboratorio de Histología Vegetal y Etnobotánica, Departamento de Biología, Universidad Nacional Autónoma de Honduras UNAH, Tegucigalpa, Honduras.
- WENIGER B, 1987-88
Encuesta TRAMIL. enda-caribe, Santo Domingo, Rep. Dominicana.
- FORGACS P, JACQUEMIN H, MORETTI C, PROVOST J, TOUCHE A, 1983
Etude phytochimique et activités biologiques de 18 plantes de la Guyane Française. Plantes Médicinales Phytothér 17(1):22-32.
- WONG K, FENG M, SAM T, TAN G, 1994
Composition of the leaf and root oils of Eryngium foetidum L. J Essent Oil Res 6(4):369-374.
- MUNSELL H, WILLIAMS LO, GUILD LP, TROESCHER CB, NIGHTINGALE G, HARRIS RS, 1950
Composition of food plants of Central America. IV: El Salvador. Food Res 15(4):263-296.
- ARBAIN D, CANNON JR, AFRIASTINI, KARTAWINATA K, DJAMAL R, BUSTARI A, DHARMA A, ROSMAWATY H, SJAFAR M, SJAIFUL, NAWFA R, KOSELA S, 1989
Survey of some West Sumatran plants for alkaloids. Econ Bot 43(1):73-78.
- HERRERA J, 1992
Determinación de parámetros farmacológicos usados en medicina tradicional popular en la Cuenca del Caribe. Departamento de Farmacología, Facultad de Salud, Universidad del Valle, Cali, Colombia.
- SARAVIA A, 1992
Estudios sobre plantas TRAMIL. Departamento de Farmacología, Universidad de San Carlos, Guatemala, Guatemala.
- GARCIA D, SAENZ T, 1995
Toxicidad aguda de algunas plantas TRAMIL. Informe TRAMIL. Farmacognosia, Facultad de Farmacia, Universidad de Sevilla, Sevilla, España.
- SAENZ MT, FERNANDEZ MA, GARCIA MD 1997
Anti-inflammatory and analgesic properties from leaves of Eryngium foetidum L. (Apiaceae). Phytother Res 11:380–383.
- SIMON O, SINGH N, 1986
Demonstration of anticonvulsant properties of an aqueous extract of spirit weed (Eryngium foetidum). Wi Med J 35(2):121-125.
- SAUVAIN M, 1989
Etude des plantes antiparasitaires du plancton des Guyanes en Amazonie : antipaludiques et antileishmanioses (Thèse de Doctorat). Université Paris-Sud, Paris, France.
- CACERES A, SAMAYOA B, 1989
Tamizaje de la actividad antibacteriana de plantas usadas en Guatemala para el tratamiento de afecciones gastrointestinales. Guatemala, Guatemala: Dirección General de Investigaciones, Universidad de San Carlos (DIGI- USAC).
- GARCIA GM, COTO MT, GONZÁLEZ CS, PAZOS L, 1996
Toxicidad sub-crónica, en ratones, del extracto acuoso de la planta entera de Eryngium foetidum. Informe TRAMIL. Laboratorio de Ensayos Biológicos LEBi, Escuela de Medicina, Universidad de Costa Rica, San Pedro, Costa Rica.
- PAZOS L, COTO T, GONZALEZ S, QUIROS S, 2004
Toxicidad oral, subcrónica en ratones, dosis repetidas, del extracto acuoso de planta entera de Eryngium foetidum. Informe TRAMIL. Laboratorio de Ensayos Biológicos LEBi, Escuela de Medicina, Universidad de Costa Rica, San Pedro, Costa Rica.