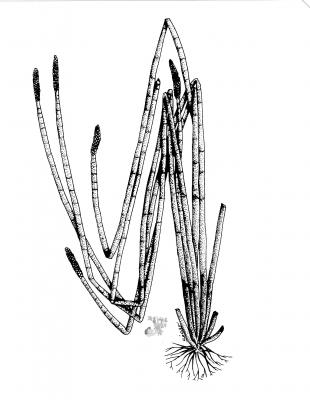
Eleocharis interstincta
Vernacular names
(In territories with significant traditional TRAMIL use)
Haiti:
- jon
Botanical description
Perennial, stoloniferous plant, with fibrous roots. Culms tufted 40-100 cm tall 3.6 mm thick, cylindrical, hollow, transversely septate; spikelets cylindrical, up to 6 cm long x 5-7 mm wide; glumes straw-coloured, oblong to obovate 4-5 mm x 3 mm; stamens 3, style 3-fid. Achenes oval to obovate 1.5-2 mm long, biconvex, slightly shiny, brownish-yellow.
Voucher(s)
Rouzier,58,FMPH
Pharmacopoeia
Ed.2References
1 WENIGER B, ROUZIER M, 1986 Enquête TRAMIL. Service Oecuménique d'Entraide SOE, Port au Prince, Haïti.
2 WHO, 1991 Pautas para la evaluación de medicamentos herbarios WHO/TRM/91.4 (original inglés). Programa de Medicina Tradicional, OMS, Ginebra, Suiza.
3 SOLIS PN, RODRIGUEZ N, ESPINOSA A, GUPTA MP, 2004 Estudio fitoquímico de algunas plantas TRAMIL con usos en Martinica. Informe TRAMIL. Centro de Investigaciones Farmacognósticas de la Flora Panameña CIFLORPAN, Facultad de Farmacia, Universidad de Panamá, Panamá, Panamá.
4 HERRERA J, 1992 Determinación de parámetros farmacológicos usados en medicina tradicional popular en la Cuenca del Caribe. Informe TRAMIL. Dep. de Farmacología, Facultad de Salud, Universidad del Valle, Cali, Colombia. TRAMIL VI, Basse Terre, Guadeloupe, UAG/enda-caribe.
5 MAGALHAES AF, RUIZ AL, FARIA AD, MAGALHAES EG, AMARAL MC, 2001 Avaliação fitoquímica e biológica de Eleocharis interstincta (Vahl) Roem. & Schult. Poços de Caldas, Brasil: Científico Nacional, Reunião Anual da Sociedade Brasileira de Química - SBQ (1):7.
6 PAZOS L, COTO T, CAIZA F, 2009
Irritación dérmica, piel sana en conejos, porción de la planta entera fresca de Eleocharis interstinca. Informe TRAMIL. Laboratorio de Ensayos Biológicos, LEBi, Universidad de Costa Rica, San Pedro, Costa Rica.