Vernacular names
(In territories with significant traditional TRAMIL use)
Barbados:
- ginger
Dominica:
- ginger
Puerto Rico:
- ginger
- jengibre
St Vincent and Grenadines:
- ginger
Saint Lucia:
- ginger
Antigua and Barbuda:
- ginger
Guatemala:
- jengibre
Honduras:
- jengibre
Venezuela:
- jengibre
Costa Rica:
- jengibre
Botanical description
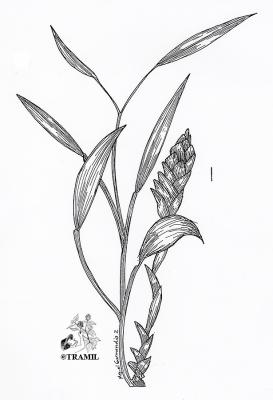
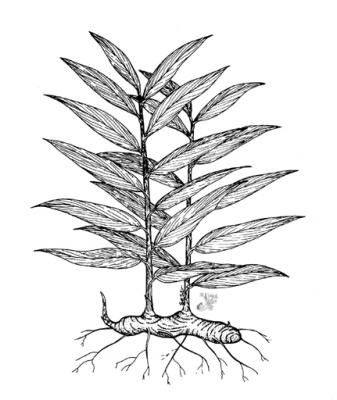
Herb 30-100 cm high, with robust, branched, rhizome near the surface of the soil. Leaves, 5-25 x 1-3 cm, linear-lanceolate, sessile, with acute apex and cuneate base, glabrous; inflorescence cylindrical, spikes 4-7 x 1.5-2.5 cm, corolla yellowish-green; flower produced in the axil of yellowish bracts; fruit a three-valved capsule with small black arillate seeds.
Voucher(s)
Faujour,4,BAR
González,37,MAPR
Girón,286,CFEH
Ochoa,315,HPMHV
Gimenez,275697-25,VEN
Balland,42,HVB
Pharmacopoeia
Ed.2References
1 DELENS M, 1990 Encuesta TRAMIL en Barlovento, Edo. Miranda de Venezuela. Centro al Servicio de la Acción Popular CESAP, Caracas, Venezuela.
2 BENEDETTI MD, 1994 Encuesta TRAMIL. Universidad de Puerto Rico, Mayagüez, Puerto Rico.
3 LAGOS-WITTE S, 1988-89, 1996 Encuesta TRAMIL. Laboratorio de Histología Vegetal y Etnobotánica, Departamento de Biología, Universidad Nacional Autónoma de Honduras UNAH, Tegucigalpa, Honduras.
4 DELENS M, 1992 Encuesta TRAMIL en los Estados Lara y Sucre de Venezuela. Centro al Servicio de la Acción Popular CESAP, Caracas, Venezuela.
5 OCAMPO R, 1988 Encuesta TRAMIL (Costa atlántica), Instituto de Desarrollo Agrario, Universidad de Costa Rica, San José, Costa Rica.
6 O'REILLY A, WILSON V, PHILLIP M, JOSEPH O, 1992 TRAMIL survey. Chemistry and Food Technology Division, Ministry of Agriculture, Dunbars, Antigua and Barbuda.
7 GERMOSEN-ROBINEAU L, GERONIMO M, AMPARO C, 1984 Encuesta TRAMIL. enda-caribe, Santo Domingo, Rep. Dominicana.
8 GIRON L, 1988 Encuesta TRAMIL (Costa atlántica). Centro Mesoamericano de Tecnología CEMAT, Guatemala, Guatemala.
9 JEAN-PIERRE L, 1988 TRAMIL survey. St. Lucia national herbarium, Castries, St. Lucia.
10 CHARLES C, 1988 TRAMIL survey. Movement for Cultural Awareness MCA, Roseau, Dominica.
11 FAUJOUR A, MURREY D, CHELTENHAM-CORBIN B, CARRINGTON S, 2003 TRAMIL survey. enda-caribbean, IICA & UAG, Saint Thomas, Barbados.
12 BALLAND V, GLASGOW A, SPRINGER F, GAYMES G, 2004 TRAMIL survey. enda-caribbean, IICA, UAG & U.PARIS XI, Saint Vincent.
13 WHO, 1991 Pautas para la evaluación de medicamentos herbarios WHO/TRM/91.4 (original inglés). Programa de Medicina Tradicional, OMS, Ginebra, Suiza.
14 WHO, 1999 Rhizoma zingiberis. WHO monographs on selected medicinal plants, Volume I. WHO: Geneva, Switzerland. p284.
15 CANIGUERAL S, 2003 Zingiber officinalis. Vademecum de Fitoterapia, Editorial Masson, Barcelona, España, Jul. 30, 2003. URL: http://www.masson.es/book/fitoterapia.html
16 TANABE M, YASUDA M, ADACHI Y, KANOY, 1991 Capillary GC-MS analysis of volatile components in Japanese gingers. Shoyakugaku Zasshi 45(4):321-326.
17 NISHIMURA O, 1995 Identification of the characteristic odorants in fresh rhizomes of ginger (Zingiber oficinale Roscoe) using aroma extract dilution analysis and modified multidimensional gas chromatography-mass spectroscopy. J Agric Food Chem 43(11):2941-2945.
18 SAKAMURA F, OGIHARA K, SUGA T, TANIGUCHI K, TANAKA R, 1986 Volatile constituents of Zingiber officinale rhizomes produced by in vitro shoot tip culture. Phytochemistry 25(6):1333-1335.
19 WU P, KUO MC, HO CT, 1990 Glycosidically bound aroma compounds in ginger (Zingiber officinale Roscoe). J Agric Food Chem 38(7):1553-1555.
20 HAGINIWA J, HARADA M, MORISHITA I, 1963 Pharmacological studies on crude drugs. VII. Properties of essential oil components of aromatics and their pharmacological effect on mouse intestine. Yakugaku Zasshi 83:624.
21 VAN BEEK TA, LELYVELD GP, 1991 Isolation and identification of the five major sesquiterpene hydrocarbons of ginger. Phytochem Anal 2(1):26-34.
22 SHIBA M, MYATA A, OKADA M, WATANABE K, 1986 Antiulcer furanogermenone extraction from ginger. Patent-Japan Kokai Tokkyo Koho-61 227,523.
23 YOSHIKAWA M, HATAKEYAMA S, CHATANI N, NISHINO Y, YAMAHARA J, 1993 Qualitative and quantitative analysis of bioactive principles in Zingiberis Rhizoma by means of high performance liquid chromatography and gas liquid chromatography. On the evaluation of Zingiberis Rhizoma and chemical change of constituents during Zingiberis Rhizoma processing. Yakugaku Zasshi 113(4):307-315.
24 TANABE M, CHEN YD, SAITO KI, KANO Y, 1993 Cholesterol biosynthesis inhibitory component from Zingiber officinale Roscoe. Chem Pharm Bull 41(4):710-713.
25 KANO Y, TANABE M, YASUDA M, 1990 On the evaluation of the preparation of Chinese medicinal prescriptions (V) diterpenes from Japanese ginger "kintoki". Shoyakugaku Zasshi 44(1):55-57.
26 KAWAKISHI S, MORIMITSU Y, OSAWA T, 1994 Chemistry of ginger components and inhibitory factors of the arachidonic acid cascade. Asc Symp Ser 547:244-250.
27 KIKUZAKI H, NAKATANI N, 1993 Antioxidant effects of some ginger constituents. J Food Sci 58(6):1407-1410.
28 KIUCHI F, IWAKAMI S, SHIBUYA M, HANAOKA F, SANKAWA U, 1992 Inhibition of prostaglandin and leukotriene biosynthesis by gingerols and diarylheptanoids. Chem Pharm Bull 40(2):387-391.
29 HARVEY DJ, 1981 Gas chromatographic and mass spectrometric studies of ginger constituents. identification of gingerdiones and new hexahydrocurcumin analogues. J Chromatogr 211(1):75-84.
30 MASADA Y, INOUE T, HASHIMOTO K, FUJIOKA M, UCHINO C, 1974 Studies on the constituents of ginger (Zingiber officinale Roscoe) by GC-MS. Yakugaku Zasshi 94(6):735-738.
31 ANON, 1982 Analgesic formulations containing shogaol and gingerol. Patent-Japan Kokai Tokkyo Koho-82 46,914.
32 CHEN CC, ROSEN RT, HO CT, 1986 Chromatographic analyses of isomeric shogaol compounds derived from isolated gingerol compounds of ginger (Zingiber officinale Roscoe). J Chromatogr 360:175-184.
33 HARTMAN M, 1971 Chemical composition of certain products from ginger (Zingiber officinale). Zivocisna Vyroba 16(10/11):805-812.
34 SCHULTZ JM, HERRMANN K, 1980 Occurrence of hydroxybenzoic acids and hydroxycinnamic acid in spices. IV. Phenolics of spices. Z Lebensm-Unters Forsch 171:193-199.
35 FU HY, HUANG TC, HO CT, DAUN H, 1993 Characterization of the major anthocyanin in acidified green ginger (Zingiber officinale Roscoe). Zhongguo Nongye Huaxue Huizhi 31(5):587-595.
36 NELSON EK, 1920 Constitution of capsaicin, the pungent principle of ginger. II. J Amer Chem Soc 42:597-599.
37 LIN ZK, HUA YF, 1987 Chemical constituents of the essential oil from Zingiber officinale Roscoe. of Sichuan. You-Ji Hua Hsueh 6:444-448.
38 ERLER J, VOSTROWSKY O, STROBEL H, KNOBLOCH K, 1988 Essential oils from ginger (Zingiber officinalis Roscoe). Z Lebensm-Unters Forsch 186(3):231-234.
39 DUKE JA, ATCHLEY AA, 1986 Handbook of proximate analysis tables of higher plants.Boca Raton, USA: CRC Press. p172.
40 KIUCHI F, SHIBUYA M, KINOSHITA T, SANKAWA U, 1983 Inhibition of prostaglandin biosynthesis by the constituents of medicinal plants. Chem Pharm Bull 31(10):3391-3396.
41 KIUCHI F, SHIBUYA M, SANKAWA U, 1982 Inhibitors of prostaglandin biosynthesis from ginger. Chem Pharm Bull 30(2):754-757.
42 SANKAWA U, 1983 Modulators of arachidonate cascade contained in medicinal plants used in traditional medicine. 3º Congress of the Federation of Asian and Oceanian biochemists, Bangkok, Thailand, p28.
43 SRIVASTAVA KC, 1984 Aqueous extracts of onion, garlic and ginger inhibited platelet aggregation and altered arachidonic acid metabolism. Biomed Biochim Acta 43(8/9):5335-5346.
44 ADACHI I, YASUTA A, MATSUBARA T, UENO M, TERASAWA K, HORIKOSHI I, 1984 Macrophage procoagulant activity. Effects of hot water extracts of several Kanpo-prescriptions on macrophage procoagulant activity, I. Yakugaku Zasshi 104(9):959-965.
45 MASCOLO N, JAIN R, JAIN SC, CAPASSO F, 1989 Ethnopharmacologic investigation of ginger (Zingiber officinale). J Ethnopharmacol 27(1/2):129-140.
46 WOO W, LEE E, HAN B, 1979 Biological evaluation of Korean medicinal plants. III. Arch Pharm Res 2:127-131.
47 MAY G, WILLUHN G, 1978 Antiviral activity of aqueous extracts from medicinal plants in tissue cultures. Arzneim-Forsch 28(1):1-7.
48 ADEWUNMI CO, 1984 Natural products as agents of schistosomiasis control in Nigeria: A review of progress. Int J Crude Drug Res 22(4):161-166.
49 FEROZ H, KHARE AK, SRIVASTAVA MC, 1982 Review of scientific studies on anthelmintics from plants. J Sci Res Pl Med 3:6-12.
50 PANTHONG A, SIVAMOGSTHAM P, 1974 Pharmacological study of the action of ginger (Zingiber officinale Roscoe) on the gastrointestinal tract. Chien Mai Med Bull 13(1):41-53.
51 KASAHARA Y, SAITO E, HIKINO H, 1983 Pharmacological actions of Pinellia tubers and Zingiber rhizomes. Shoyakugaku Zasshi 37(1):73-83.
52 SAKAI K, MIYAZAKI Y, YAMANE T, SAITOH Y, IKAWA C, NISHIHATA T, 1989 Effect of extracts of Zingiberaceae herbs on gastric secretion in rabbits. Chem Pharm Bull 37(1):215-217.
53 YAMAHARA J, RONG HQ, NAITOH Y, KITANI T, FUJIMURA H, 1989 Inhibition of cytotoxic drug-induced vomiting in Suncus by a ginger constituent. J Ethnopharmacol 27(3):353-355.
54 YAMAHARA J, YAMADA T, KIMURA H, SAWADA T, FUJIMURA H, 1982 Biologically active principles of crude drugs. Anti-allergic principles of "Shoseiryu-To". I. Effect on delayed-type allergy reaction. Yakugaku Zasshi 102(9):881-886.
55 MOWREY DB, CLAYSON DE, 1982 Motion sickness, ginger and psychophysics. Lancet 82(1):655-657.
56 GRONTVED A, BRASK T, KAMBSKARD J, HENTZER E, 1988 Ginger root against seasickness. A controlled trial on the open sea. Acta Otolaryngol (Stockholm) 105(1/2):45-49.
57 HOLTMANN S, CLARKE AH, SCHERER H, HOHN M, 1989 The anti-motion sickness mechanism of ginger. A comparative study with placebo and dimenhydrinate. Acta Otolaryngol (Stockholm) 108(3/4):168-174.
58 WOOD CD, MANNO JE, WOOD MJ, MANNO BR, MIMS ME, 1988 Comparison of efficacy of Ginger with various antimotion sickness drug. Clin Res Pract Drug Reg Affairs 6(2):129-136.
59 FISCHER-RASMUSSEN W, KJAER SK, DAHL C, ASPING U, 1991 Ginger treatment of hyperemesis gravidarum. Eur J Obstetr Gynecol Reprod Biol 38(1):19-24.
60 SRIVASTAVA KC, 1989 Effect of onion and ginger consumption on platelet thromboxane production in humans. Prostaglandins Leukotrienes Essent Fatty Acids 35(3):183-185.
61 BETANCOURT J, MARTINEZ MJ, LOPEZ M, MOREJON Z, BARCELO H, LAINEZ A, MONTES ME, REGO R, BOUCOURT E, MORON F, 2000 Toxicidad aguda clásica de rhizome de Zingiber officinalis Roscoe. Laboratorio Central de Farmacología, Facultad de Ciencias Médicas “Dr. Salvador Allende”, La Habana, Cuba.
62 BETANCOURT J, MARTINEZ MJ, LOPEZ M, MOREJON Z, BOUCOURT E, MORON F, 2000 Actividad genotóxica in vitro de rhizome de Zingiber officinalis Roscoe. Laboratorio Central de Farmacología, Facultad de Ciencias Médicas “Dr. Salvador Allende”, La Habana.
63 CARBALLO A, 1995 Plantas medicinales del Escambray cubano. Informe TRAMIL. Laboratorio provincial de producción de medicamentos, Sancti Spiritus, Cuba.
64 ASWAL BS, BHAKUNI DS, GOEL AK, KAR K, MEHROTRA BN, MUKHERJEE KC, 1984 Screening of Indian plants for biological activity: Part X. Indian J Exp Biol 22(6):312-332.
65 EMIG H, 1931 The pharmacological action of ginger. J Amer Pharm Ass 20:114-116.
66 ANON (Select Committee on GRAS Substances), 1976 GRAS status of foods and food additives. Washington DC, USA: Food and Drug Administration, Department of Health and Human Services, Office of the Federal Register National Archives and Records Administration 41, 38644
67 KUMAZAWA Y, TAKIMOTO H, MIURA SI, NISHIMURA C, YAMADA A, KAWAKITA T, NOMOTO K, 1988 Activation of murine peritoneal macrophages by intraperitoneal administration of a traditional Chinese herbal medicine, Xiao-Chai-Hu-Tang (Japanese name: Shosaiko-To). Int J Inmunopharmacol 10(4):395-403.
68 PERIS JB, STUBING G, 2003 Zingiber officinalis. Vademecum de Fitoterapia, Editorial Masson, Barcelona, España, Jul. 30, 2003. URL: http://www.masson.es/book/fitoterapia.html