Vernacular names
(In territories with significant traditional TRAMIL use)
Martinique:
- brisée
- lamerik
Venezuela:
- malojillo extranjero
- poleo
- Santa María
- toronjil
Panama:
- mastranto
Puerto Rico:
- poleo
Saint Martin:
- ti bouch
Geographical distribution
Tropical and subtropical America.
Botanical description
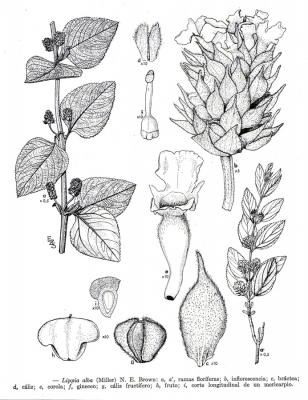
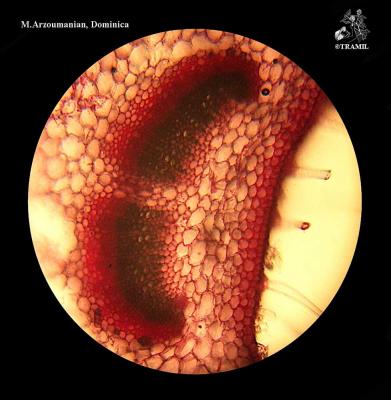
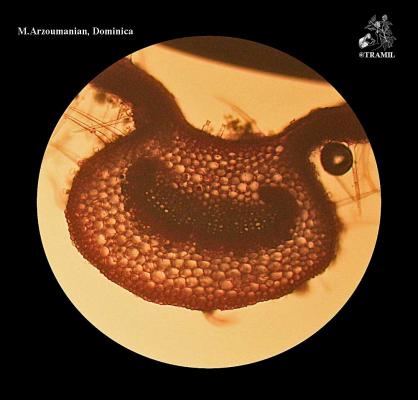
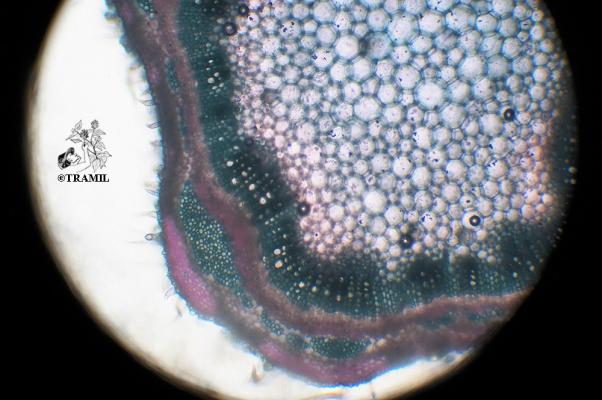
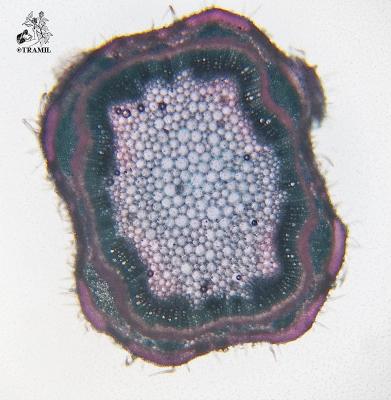
Aromatic shrub, 1 to 2 m high, long wicker branches, decumbent and rooting, densely puberulent or strigose.
Opposite or ternate leaves, briefly petiolate, 2-6 (rarely 9) cm in length and 1.2-4.5 cm wide, oblong, wrinkled, scalloped, covered with short, small hairs and prominent venation. Inflorescences in globular axillary heads, cylindrical in fructification. Bracts ovate, acuminate, lower bracts mucronate. Zygomorphic flowers, calyx 1.5-1.7 mm long and accrescent at maturity; purple corolla with yellow and white jaws, pink or white 4 to 5 mm long. 3 mm wide obovoid fruit.
Voucher(s)
Gimenez,34,VEN
FLORPAN,1933,PMA
Longuefosse&Nossin,65,HAVPM
Ocrisse,2,UAG
Pharmacopoeia
Ed.3References
1 OCRISSE G, 2008
Enquête TRAMIL auprès de 211 familles du canton Est de la partie francophone de St Martin. Biologie végétale, UAG, Guadeloupe (FWI),
2 LONGUEFOSSE JL, NOSSIN E, 1990-95
Enquête TRAMIL. Association pour la valorisation des plantes médicinales de la Caraïbe AVPMC, Fort de France, Martinique.
3 DELENS M, 1992
Encuesta TRAMIL en los Estados Lara y Sucre de Venezuela. Centro al Servicio de la Acción Popular CESAP, Caracas, Venezuela.
4 BENEDETTI MD, MEJIA A, ACOSTA D, 1994
Encuesta TRAMIL. Universidad de Puerto Rico, Mayagüez, Puerto Rico.
5 SOLIS P, CORREA M, GUPTA M, 1995
Encuesta TRAMIL (Comunidades afro-caribeñas). Centro de Investigaciones Farmacognósticas de la Flora Panameña CIFLORPAN, Facultad de Farmacia, Universidad de Panamá, Panamá, Panamá.
6 CATALAN CAN, MEREP DJ, RETAMAR JA, 1977
The essential oil of Lippia alba from the Tucuman province. Riv Ital Essenze Profumi Piante Offic Aromi Saponi Cosmet Aer 59:513-518.
7 FESTER G, MARTINUZZI E, RETAMAR J, RICCIARDI A, 1955
Some volatile essential oils. VII. Rev Fac Ing Quim 24:37-55.
8 CASTRO DM, MING LC, MARQUES MO, TANAKA FA, 2000
Anatomic characterization and chemical composition of essential oils of different leaves of Lippia alba (Mill.) stem axis. 1er Congreso Peruano de Plantas Medicinales y Fitoterapía, 27-30 Septiembre 2000, Lima, Perú:112-114.
9 BANDONI A, 2003
Los recursos vegetales aromáticos en Latinoamérica: su aprovechamiento industrial para la producción de aromas y sabores. Buenos Aires, Argentina: CYTED, 2 ed.,
CDROM, ISBN:987-43-6072-0.
10 LÓPEZ MA, STASHENKO EE, FUENTES JL, 2011
Chemical composition and antigenotoxic properties of Lippia alba essential oils. Genet Mol Biol 34(3):479-488.
11 VERAS HN, CAMPOS AR, RODRIGUES FF, BOTELHO MA, COUTINHO HD, MENEZES IR, DA COSTA JG, 2011
Enhancement of the antibiotic activity of erythromycin by volatile compounds of Lippia alba (Mill.) N.E. Brown against Staphylococcus aureus. Pharmacogn Mag 7(28):334-337.
12 MAYNARD LG, SANTOS KC, CUNHA PS, BARRETO AS, PEIXOTO MG, ARRIGONIBLANK F, BLANK AF, ALVES PB, BONJARDIN LR, SANTOS MR, 2011
Chemical composition and vasorelaxant effect induced by the essential oil of Lippia alba (Mill.) N.E. Brown. (Verbenaceae) in rat mesenteric artery. Indian J Pharmacol
43(6):694-698.
13 CONDE R, CORRÊA VS, CARMONA F, CONTINI SH, PEREIRA AM, 2011
Chemical composition and therapeutic effects of Lippia alba (Mill.) N. E. Brown leaves hydro-alcoholic extract in patients with migraine. Phytomedicine 18(14):1197-1201.
14 BANDONI AL, MENDIONDO ME, RONDINA RVD, COUSSIO JD, 1976
Survey of Argentine medicinal plants. Folklore and phytochemical screening. II. Econ Bot 30(2):161-185.
15 RUFFA MJ, WAGNER ML, SURIANO M, VICENTE C, NADINIC J, PAMPURO S, SALOMON H, CAMPOS RH, CAVALLARO L, 2004
Inhibitory effect of medicinal herbs against RNA and DNA viruses. Antivir Chem Chemother 15(3):153-159.
16 CACERES A, ALVAREZ AV, OVANDO AE, SAMAYOA BE, 1991
Plants used in Guatemala for the treatment of respiratory diseases. 1. Screening of 68 plants against gram-positive bacteria. J Ethnopharmacol 31(2):193-208.
17 COSTA M, DI STASI L, KIRIZAWA M, MENDACOLLI S, GOMES C, TROLIN G, 1989
Screening in mice of some medicinal plants used for analgesic purposes in the state of Sao Paulo. J Ethnopharmacol 27(1/2):25-33.
18 VALE TG, MATOS FJA, DE LIMA TCM, VIANA GSB, 1999
Behavioral effects of essential oils from Lippia alba (Mill.) N.E. Brown chemotypes. J Ethnopharmacol 67(2):127-133.
19 GARCIA GM, COTO MT, GONZALEZ CS, OCAMPO R, PAZOS L, 2001
Toxicidad aguda en ratones, del extracto acuoso de hoja fresca de Lippia alba. Informe TRAMIL. Laboratorio de Ensayos Biológicos LEBi, Escuela de Medicina, Universidad de Costa Rica, San Pedro, Costa Rica.
20 PAZOS L, COTO T, GONZALEZ S, 2003
Toxicidad oral, aguda en ratón, del extracto acuoso de partes aéreas de Lippia alba var. dulce. Informe TRAMIL. Laboratorio de Ensayos Biológicos LEBi, Universidad de Costa Rica, San Pedro, Costa Rica.
21 PAZOS L, COTO T, GONZALEZ S, QUIROS S, 2004
Toxicidad oral, aguda en ratón, dosis repetidas, del extracto acuoso de partes aéreas de Lippia alba. Informe TRAMIL. Laboratorio de Ensayos Biológicos LEBi, Universidad de Costa Rica, San Pedro, Costa Rica.
22 PASCUAL ME, SLOWING K, CARRETERO ME, VILLAR A, 2001
Antiulcerogenic activity of Lippia alba (Mill.) N. E. Brown (Verbenaceae). Farmaco 56(5-7):501-504.
23 BHAKUNI D, DHAR M, DHAR M, DHAWAN B, GUPTA B, SRIMALI R, 1971
Screening of Indian plants for biological activity. Part III. Indian J Exp Biol 9:91.
24 CARBALLO A, 1994
Plantas medicinales del Escambray Cubano. Apuntes científicos.
25 Code of Federal Regulations, 2004
Food and drugs. Chapter I - Food and Drug Administration, Department of Health and Human services. Part 182 - Substances generally recognized as safe. Sec. 182.10.
Spices and other natural seasonings and flavorings. U.S. Government Printing Office via GPO Access, USA. 21(3):451-452. Nov.13,2004, URL: www.cfsan.fda.gov/~lrd/FCF182.html