Vernacular names
(In territories with significant traditional TRAMIL use)
Haiti:
- chapantyé
Cuba:
- tilo
Costa Rica:
- tilo
Martinique:
- zèb chapantyé
Dominica:
- zèb chapantyé
Other vernacular names
Dominica:
- Carpenter's grass
- Carpenter's grass o Z'herbe carpentier
- Carpenter's grass o Zeb chepentier
French Guiana:
- Crementin
Puerto Rico:
- Curía
Costa Rica:
- Flor de tilo
Guadeloupe:
- herbe charpentier
Haiti:
- Sepantye
- Zeb chapantye
Cuba:
- Tilo
- Tilo
Martinique:
- zèb chapantyé, herbe charpentier
Botanical description
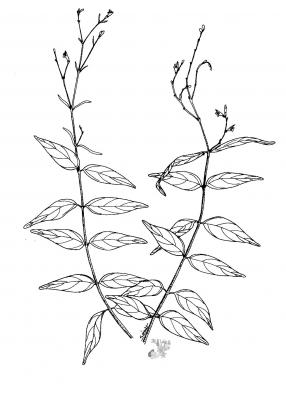
Perennial low branching herb, aromatic when crushed, up to 2 m high, sometimes rooting at the nodes. Leaves opposite, 3-10 cm x 5-30 mm, linear to lanceolate; inflorescence terminal in loose panicle; flowers 7-8 mm long, pink with white markings resembling a rib-cage; capsule dehiscing elastically.
Voucher(s)
Ocampo,5000,CR Soberats,TR90-02,CIFMT Longuefosse&Nossin,21,HAVPMC Rouzier,220,SOE Fuentes,4758,ROIG
Pharmacopoeia
Ed.2References
1 WENIGER B, ROUZIER M, 1986 Enquête TRAMIL. Service Oecuménique d'Entraide SOE, Port au Prince, Haïti.
2 CHARLES C, 1988 TRAMIL survey. Movement for Cultural Awareness MCA, Roseau, Dominica.
3 LONGUEFOSSE JL, NOSSIN E, 1990-95 Enquête TRAMIL. Association pour la valorisation des plantes médicinales de la Caraïbe AVPMC, Fort de France, Martinique.
4 CARBALLO A, 1990 Encuesta TRAMIL. Centro de investigación de fitoterapia y medicina tradicional de Topes de Collantes, Trinidad, Cuba.
5 OCAMPO R, 1988 Encuesta TRAMIL (Costa atlántica), Instituto de Desarrollo Agrario, Universidad de Costa Rica, San José, Costa Rica.
6 WHO, 1991 Pautas para la evaluación de medicamentos herbarios WHO/TRM/91.4 (original inglés). Programa de Medicina Tradicional, OMS, Ginebra, Suiza.
7 Solis PN, VAsquez Y, Ayala H, Gupta MP, 2002 Informe de validación de algunas plantas tramil. Fase iii. Informe TRAMIL. Centro de Investigaciones Farmacognósticas de la Flora Panameña CIFLORPAN, Facultad de Farmacia, Universidad de Panamá, Panamá, Panamá.
8 WENIGER B, SAVARY H, DAGUIHL R, 1984 Tri phytochimique de plantes de la liste TRAMIL. Laboratoire de chimie des substances naturelles, Faculté de Médecine et de Pharmacie, Université d'Etat d'Haïti, Port au Prince, Haïti.
9 DE VRIES JX, TAUSCHER B, WURZEL G, 1988 Constituents of Justicia pectoralis Jacq. 2. Gas chromatography/mass spectrometry of simple coumarins, 3-phenylpropionic acids and their hydroxy and methoxy derivatives. Biomed Environ Mass Spectrom 15(8):413-417.
10 JOSEPH H, GLEYE J, MOULIS C, MENSAH L, ROUSSAKIS C, GRATAS C, 1988 Justicidin B, a cytotoxic principle from Justicia pectoralis. J Nat Prod 51(3):599-600.
11 DUKE JA, ATCHLEY AA, 1986 Handbook of proximate analysis tables of higher plants.Boca Raton, USA: CRC Press, p90.
12 GarcIa GM, Coto MT, GonzAlez CS, Pazos L, 1998 Velocidad del tránsito intestinal en ratón, del extracto acuoso de hoja fresca de Justicia pectoralis. Informe TRAMIL. Laboratorio de Ensayos Biológicos LEBI, Escuela de Medicina, Universidad de Costa Rica, San Pedro, Costa Rica.
13 CACERES A, 2000 Actividad de Justicia pectoralis contra las bacterias causales de infecciones respiratorias. Informe TRAMIL.Facultad de Ciencias Químicas y Farmacia, Universidad de San Carlos USAC, Guatemala, Guatemala.
14 PAZOS L, COTO T, GONZALEZ S, 2003 Actividad sedante-tranquilizante, en ratones, del extracto acuoso de partes aéreas de Justicia pectoralis. Informe TRAMIL. Laboratorio de Ensayos Biológicos, Universidad de Costa Rica, San Pedro, Costa Rica.
15 MACRAE W, TOWERS G, 1984 Justicia pectoralis: A study of the basis for its use as a hallucinogenic snuff ingredient. J Ethnopharmacol 12(1):93-111.
16 FernAndez L, PErez H, Mas R, RodrIguez L, GalAn L, Biscay R, 1987 Efecto de Justicia pectoralis sobre la conducta exploratoria en ratones. Centro Nacional de Investigaciones Científicas. Estudios Avanzados en Neurociencias (suppl 1). p257-264.
17 FERNANDEZ L, PEREZ SAAD H, MAS R, RODRIGUEZ RIVERA L, GALAN L, BISCAY R, 1987 Efecto de Justicia pectoralis sobre la conducta exploratoria en ratones. En: Centro Nacional de Investigaciones Científicas (CENIC) Ed. Estudios avanzados en neurociencias. La Habana, Cuba: Editorial CENIC. p257-264.
18 FERNANDEZ L, MAS R, PEREZ SAAD H, BISCAY R, GALAN L, 1989 Evaluación preliminar de los efectos neurofarmacológicos de Justicia pectoris. Rev Cub Farm 23(1/2):161-166.
19 RODRIGUEZ E, VIRNES A, ALEMAN J, 1989 Estudio preliminar del efecto de Justicia pectoralis sobre el EEG de adultos normales. Rev Cub Farm 23(3):302-308.
20 MAS R; MENENDEZ R, FERNANDEZ L, PEREZ SAAD H, RODRIGUEZ RIVERA L, KAMMERER E, 1987 ¿Posee Justicia pectoralis las características farmacológicas de los neurolépticos clásicos? En: Centro Nacional de Investigaciones Científicas (CENIC) Ed. Estudios avanzados en neurociencias. La Habana, Cuba: Editorial CENIC. p273-283.
21PEREZ SAAD H, MAS R, FERNANDEZ L, RODRIGUEZ RIVERA L, 1987 Justicia pectoralis no previene las convulsiones inducidas por PTZ y PTX. En: Centro Nacional de Investigaciones Científicas (CENIC) Ed. Estudios avanzados en neurociencias. La Habana, Cuba: Editorial CENIC. p265-272.
22 MILLS J, Pascoe KO, Chambers J, Melville GN, 1986 Preliminary investigations of the wound-healing properties of a Jamaican folk medicine plant. West Indian Med J 35(3):190-193.
23 GarcIa GM, Coto MT, OCAMPO R, GonzAlez CS, Pazos L, 2001 Toxicidad aguda en ratones del extracto acuoso de partes aéreas de Justicia pectoralis. Informe TRAMIL. Laboratorio de Ensayos Biológicos LEBI, Escuela de Medicina, Universidad de Costa Rica, San Pedro, Costa Rica.
24 MARTINEZ MJ, LOPEZ M, BETANCOURT J, FUENTES V, MOREJON Z, MORON, F, BOUCOURT E, 2002 Toxicidad aguda tópica de Justicia pectoralis Jacq. Informe TRAMIL. Facultad de Medicina "Dr. Salvador Allende". Laboratorio Central de Farmacología. La Habana, Cuba.
25 MARTINEZ MJ, LOPEZ M, BETANCOURT J, FUENTES V, MOREJON Z, MORON, F, BOUCOURT E, 2002 Irritabilidad dérmica primaria de Justicia pectoralis Jacq. Informe TRAMIL. Facultad de Medicina "Dr. Salvador Allende". Laboratorio Central de Farmacología. La Habana, Cuba.
26 PILOTO FERRER J, VIZOSO A, RAMOS A, GARCIA A, REMIGIO A, VEGA Y, GONZALEZ ML, RODRIGUEZ C, CARBALLO C, 2009 Plantas medicinales. Diez años de evaluaciones toxicogenéticas en el CIDEM. Boletín Latinoamericano y del Caribe de Plantas Medicinales y Aromáticas, 8(5):428-434.
27 MOREJON Z, LOPEZ M, GARCIA MJ, BOUCOURT E, VICTORIA M, FUENTES V, MORON F, BOULOGNE I, ROBINEAU L, 2009 Encuesta TRAMIL preliminar a grupos de vecinos en los municipios 10 de Octubre, Lisa, Marianao, Habana del Este (Cojímar) en la Ciudad de la Habana. Laboratorio Central de Farmacología, Universidad de Ciencias Médicas de La Habana, Ciudad de La Habana, Cuba.