Vernacular names
(In territories with significant traditional TRAMIL use)
Haiti:
- bwa donm
Dominican Republic:
- bwa donm
- guácima
Geographical distribution
Tropical regions of the American continent.
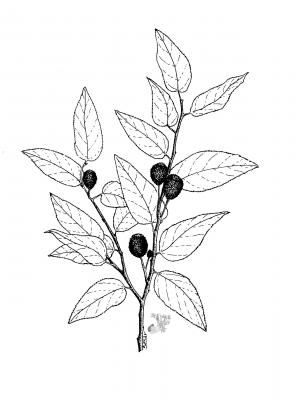
Botanical description
Shrub or tree of up to 20 m Leaves oblong to broadly ovate, of 3-15 cm, acute to acuminate, serrate, stellate-tomentose, markedly symmetrical. Fragrant, yellowish flowers in small axillary clusters, with 5 petals of 3 mm. Woody, globular or oval fruit, measuring 2-4 cm, with hard tubers.
Voucher(s)
Rouzier,192,SOE
Pimentel,1164,JBSD
Pharmacopoeia
Ed.3References
1 WENIGER B, ROUZIER M, 1986
Enquête TRAMIL. Service Oecuménique d’Entraide SOE, Port au Prince, Haïti.
2 WENIGER B, 1987-88
Encuesta TRAMIL. enda-caribe, Santo Domingo, Rep. Dominicana.
3 WENIGER B, SAVARY H, DAGUIHL R, 1984
Tri phytochimique de plantes de la liste TRAMIL. Laboratoire de chimie des substances naturelles, Faculté de Médecine et de Pharmacie, Université d’Etat d’Haïti, Port au Prince, Haïti.
4 WONG W, 1976
Some folk medicinal plants from Trinidad. Econ Bot 30:103-142.
5 SEIGLER DS, PAULI GF, FROHLICH R, WEGELIUS E, NAHRSTEDT A, GLANDER KE, EBINGER JE, 2005
Cyanogenic glycosides and menisdaurin from Guazuma ulmifolia, Ostrya virginiana, Tiquilia plicata, and Tiquilia canescens. Phytochemistry 66(13):1567-1580.
6 HOR M, HEINRICH M, RIMPLER H, 1996
Proanthocyanidin polymers with antisecretory activity and proanthocyanidin oligomers from Guazuma ulmifolia bark. Phytochemistry 42(1):109-119.
7 LOPES GC, LONGHINI R, DOS SANTOS PVP, ARAÚJO AAS, BRUSCHI ML, DE MELLO JCP, 2012
Preliminary assessment of the chemical stability of dried extracts from Guazuma ulmifolia Lam. (Sterculiaceae). Int J Anal Chem 2012:508945.
8 GIRON L, 1988
Evaluación de la actividad antibacteriana de 4 plantas de la lista TRAMIL. Facultad de Ciencias Químicas y Farmacia, Universidad de San Carlos USAC, Guatemala, Guatemala.
9 CACERES A, GONZALEZ S, GIRON L, 1998
Demostración de la actividad antimicrobiana de plantas TRAMIL en base a los usos populares en la cuenca del Caribe. Laboratorio de productos fitofarmacéuticos Farmaya y Facultad de Ciencias Químicas y Farmacia, Universidad de San Carlos, Guatemala, Guatemala.
10 CARBALLO A, 1995
Plantas medicinales del Escambray cubano. Informe TRAMIL. Laboratorio provincial de producción de medicamentos, Sancti Spiritus, Cuba.
11 TSENG C, IWAKAMI S, MIKAJIRI A, SHIBUYA M, HANAOKA F, EBIZUKA Y, PADMAWINATA K, SANKAWA U, 1992
Inhibition of in vitro prostaglandin and leukotriene biosyntheses by cinnamoyl-beta-phenethylamine and N-acyldopamine derivatives. Chem Pharm Bull 40(2):396-400.
12 NEGWER M, 1987
Organic chemical drugs and their synonyms (an international survey). 6o ed. Berlin, Germany: Akademie Verlag.
13 DUKE JA, 1992
Handbook of biologically active phytochemicals and their bioactivities. Boca Raton, USA: CRC Press.
14 MARTINEZ MJ, LOPEZ M, MOREJON Z, BOUCOURT E, FUENTES V, MORON F, 2007
Irritabilidad dérmica primaria de corteza del tallo fresco machacado de Guazuma ulmifolia Lam. Informe TRAMIL. Laboratorio Central de Farmacología, Facultad de Ciencias Médicas “Dr. Salvador Allende”, La Habana, Cuba.
15 MARTINEZ MJ, LOPEZ M, MOREJON Z, BOUCOURT E, FUENTES V, MORON F, 2007
Irritabilidad dérmica primaria de hoja seca machacada de Guazuma ulmifolia Lam. Informe TRAMIL. Laboratorio Central de Farmacología, Facultad de Ciencias Médicas “Dr. Salvador Allende”, La Habana, Cuba.
16 HERRERA J, 1990
Determinación de parámetros farmacológicos en vegetales utilizados en medicina tradicional en la Cuenca del Caribe. Informe TRAMIL. Laboratorio de fitofarmacología, Departamento de Farmacología, Facultad de Salud, Universidad del Valle, Cali, Colombia.
17 HOEHNE FC, 1939
Plantas e substâncias vegetais tóxicas e medicinais. São Paulo, Brazil: Dep. Bot. do Estado Sao-Paulo. Ed Graphicars.
18 CARBALLO A, 1995
Cálculo de concentración y dosis de las drogas vegetales TRAMIL: Mensuraciones farmacognósticas y aproximaciones técnico- clínicas. Laboratorio provincial de producción de medicamentos, Sancti Spiritus, Cuba.
19 CACERES A, 1996
Plantas de uso medicinal en Guatemala. Guatemala, Guatemala: Editorial Universitaria de San Carlos. p126.