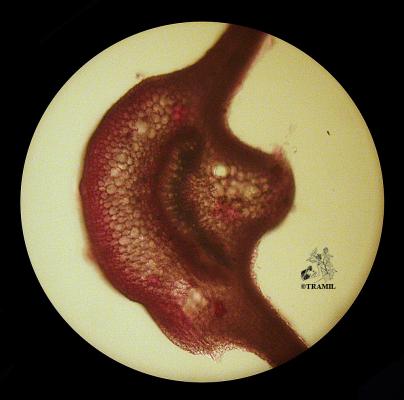
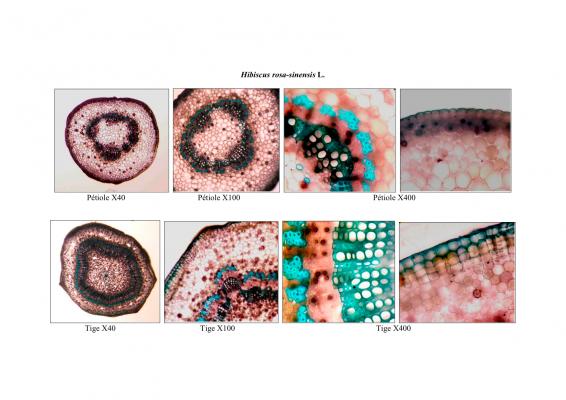
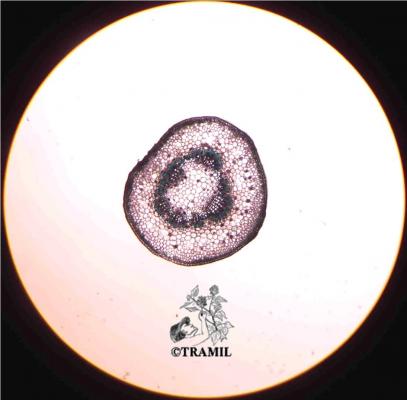
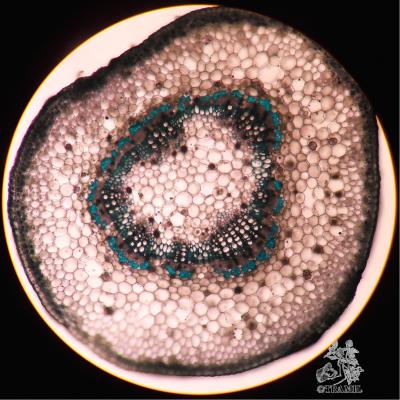
Vernacular names
(In territories with significant traditional TRAMIL use)
Haiti:
- choublak
Martinique:
- kokliko wouj
Guadeloupe:
- rose kayenn
Geographical distribution
Native to Asia, cultivated in tropical areas of the world.
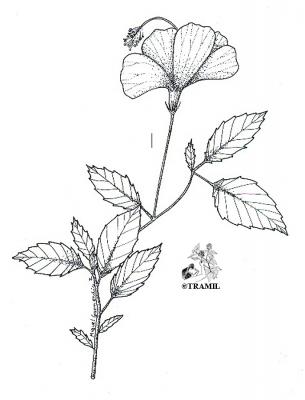
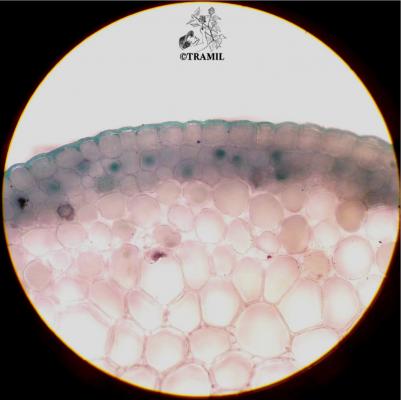
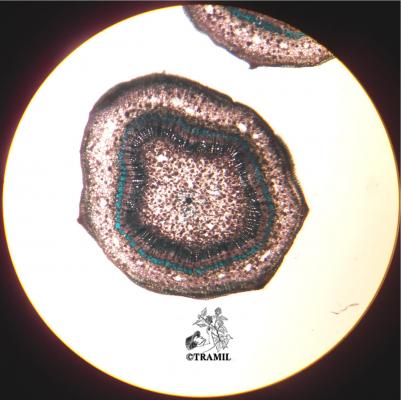
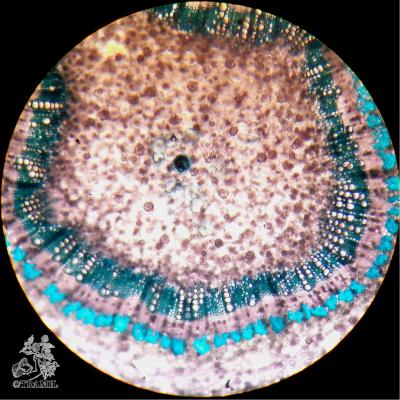
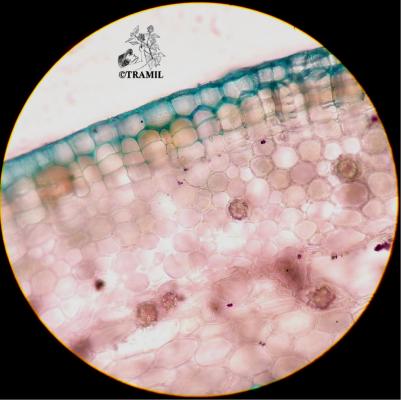
Botanical description
Shrub of 1 to 4 m in height. Alternate leaves, ovate, sharp to acuminate in the apex, serrate. 5 to 7 bracteoles, linear. Campanulate calyx; white, red-purple, yellow or orange corolla, variable in size, very bright and colourful; staminal tube longer than petals. Oblong capsule almost 3 cm in diameter.
Voucher(s)
Rouzier,252,SOE
Longuefosse&Nossin,19,HAVPM
Martínez,4739,ROIG
Boulogne,TH,11,UAG
Boulogne,TB,9,UAGMartínez,4739,ROIG
Pharmacopoeia
Ed.2References
1 WENIGER B, ROUZIER M, 1986
Enquête TRAMIL. Service Oecuménique d’Entraide SOE, Port au Prince, Haïti.
2 SOLIS PN, ESPINOSA A, DE GRACIA J, MARTINEZ L, GUPTA MP, 2003
Encuesta TRAMIL-GEF Emberá-Wounaann. Centro de Investigaciones Farmacognósticas de la Flora Panameña, Facultad de Farmacia, Universidad de Panamá, Panamá, Panamá.
3 LONGUEFOSSE JL, NOSSIN E, 1990-95
Enquête TRAMIL. Association pour la valorisation des plantes médicinales de la Caraïbe AVPMC, Fort de France, Martinique.
4 EDOUARD JA, 1992
Enquête TRAMIL. Lycée agricole, Baie-Mahault, Guadeloupe.
5 BOULOGNE I, 2009
Enquête TRAMIL, (Terre-de-Bas et Terre-de-Haut) Les Saintes, UAG, Guadeloupe.
6 MEDITSCH J, BARROS E, 1978
Hibiscus dyes as acid-base indicators. An Assoc Bras Quim 29(1):89.
7 SHRIVASTAVA D, 1974
Phytochemical analysis of japakusum. J Res Indian Med Yoga Homeopathy 9(4):103-
104.
8 LIN Y, 1975
The study of red pigments in Taiwan plants. Proc Natl Sci Counc Part I (Taiwan) 1975(8):133-137.
9 PATTANAIK S, 1949
A comparative study of the catalase activity of the petals and leaves of Hibiscus rosa-sinensis. Curr Sci 18:212-213.
10 GRIFFITHS L, 1959
On the distribution of gentisic acid in green plants. J Exp Bot 10(3):437-442.
11 AGARWAL SL, SHINDE S, 1967
Studies on Hibiscus rosa-sinensis II. Preliminary pharmacological investigations. Indian J Med Res 55(9):1007-1010.
12 SRIVASTAVA DN, BHATT SK, UDUPA KN, 1976
Gas chromatographic identification of fatty acids, fatty alcohols, and hydrocarbons of Hibiscus rosa-sinensis leaves. J Amer Oil Chem Soc 53:607-608.
13 DUKE JA, ATCHLEY AA, 1986
Handbook of proximate analysis tables of higher plants. Boca Raton, USA: CRC Press. p84.
14 MORON FJ, VICTORIA MdC, MARTINEZ I, BRITO G, MOREJON Z, ACOSTA L, FUENTES V, 2011
Efecto antipirético de la decocción 30% de flores frescas de Hibiscus rosa sinensis (mar Pacífico) en ratas. Informe TRAMIL. Facultad de Ciencias Médicas “Dr. Salvador Allende”, Laboratorio Central de Farmacología, La Habana, Cuba.
15 VICTORIA MdC, MORON FJ, MARTINEZ I, BRITO G, MOREJON Z, ACOSTA L, FUENTES V, 2011
Ausencia de efecto antipirético de la decocción 30% de hojas frescas de Hibiscus rosa sinensis (mar Pacífico) en ratas. Informe TRAMIL. Facultad de Ciencias Médicas “Dr. Salvador Allende”, Laboratorio Central de Farmacología, La Habana, Cuba.
16 VICTORIA MC, MORON FJ, MARTINEZ I, BRITO G, MOREJON Z, ACOSTA L, FUENTES V, 2011
Efecto analgésico oral de la decocción 30% de flores frescas de Hibiscus rosa sinensis (mar Pacífico) en ratones. Informe TRAMIL. Facultad de Ciencias Médicas “Dr. Salvador Allende”, Laboratorio Central de Farmacología, La Habana, Cuba.
17 HERRERA J, 1994
Determinación de actividades biológicas de vegetales utilizados en medicina tradicional. Informe TRAMIL. Dep. de Farmacología, Facultad de Salud, Universidad del Valle, Cali, Colombia.
18 BHAKUNI DS, DHAR ML, DHAR MM, DHAWAN BN, MEHROTRA BN, 1969
Screening of Indian plants for biological activity. Part II. Indian J Exp Biol 7(4):250-262.
19 SINGH N, NATH R, AGARWAL AK, KOHLI RP, 1978
A pharmacological investigation of some indigenous drugs of plant origin for evaluation of their antipyretic, analgesic and anti- inflammatory activities. J Res Indian Med Yoga Homeopathy 13(2):58-70.
20 KHOLKUTE SD, CHATTERJEE S, UDUPA KN, 1976
Effect of Hibiscus rosa-sinensis on estrous cycle and reproductive organs in rats. Indian J Exp Biol 14:703-704.
21 PRAKASH A, 1979
Acid and alkaline phosphatase activity in the uterus of rat treated with Hibiscus rosa-sinensis Linn. extracts. Curr Sci 48:501- 503.
22 LOPEZ M, MARTINEZ MJ, MOREJON Z, BOUCOURT E, FERRADA C, FUENTES V, MORON F, 2005
Irritabilidad dérmica primaria de hoja fresca de Hibiscus rosa-sinensis L. Informe TRAMIL. Laboratorio Central de Farmacología, Facultad de Medicina “Dr. Salvador Allende”, Cerro, C. Habana, Cuba.
23 SINGH M, SINGH R, UDUPA K, 1982
Antifertility activity of a benzene extract of Hibiscus rosa-sinensis flowers on female albino rats. Planta Med 44:171-174.
24 PRAKASH A, 1984
Biological evaluation of some medicinal plant extracts for contraceptive efficacy. Contracept Deliv Syst 5(3):9-10.
25 TIWARI P, 1974
Preliminary clinical trial on flowers of Hibiscus rosa-sinensis as an oral contraceptive agent. J Res Indian Med Yoga Homeopathy 9(4):96-98.
26 KHOLKUTE S, UDUPA K, 1974
Antifertility properties of Hibiscus rosa-sinensis. J Res Indian Med Yoga Homeopathy 9(4):99-102.
27 TRIVEDI V, SHUKLA K, 1980
A study of effects of an indigenous compound drug on reproductive physiology. J Sci Res Pl Med 1(3/4):41-47.